Filter News
Area of Research
- (-) Materials (19)
- (-) Neutron Science (6)
- (-) Nuclear Science and Technology (1)
- Advanced Manufacturing (13)
- Biological Systems (1)
- Biology and Environment (32)
- Building Technologies (3)
- Clean Energy (104)
- Computational Biology (1)
- Computational Engineering (1)
- Computer Science (6)
- Electricity and Smart Grid (1)
- Energy Sciences (2)
- Fusion and Fission (3)
- Fusion Energy (2)
- Isotopes (4)
- Materials for Computing (4)
- National Security (7)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (7)
- Transportation Systems (2)
News Type
News Topics
- (-) 3-D Printing/Advanced Manufacturing (6)
- (-) Bioenergy (3)
- (-) Biomedical (5)
- (-) Energy Storage (10)
- (-) Sustainable Energy (3)
- (-) Transportation (8)
- Advanced Reactors (6)
- Artificial Intelligence (1)
- Buildings (1)
- Chemical Sciences (7)
- Clean Water (1)
- Composites (4)
- Computer Science (1)
- Coronavirus (1)
- Critical Materials (5)
- Decarbonization (1)
- Environment (2)
- Fusion (5)
- Isotopes (3)
- Materials (17)
- Materials Science (26)
- Microscopy (9)
- Molten Salt (4)
- Nanotechnology (13)
- Neutron Science (31)
- Nuclear Energy (18)
- Physics (9)
- Polymers (8)
- Quantum Computing (2)
- Quantum Science (2)
- Space Exploration (4)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.
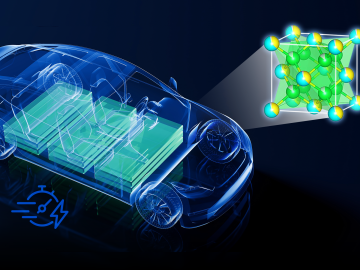
Currently, the biggest hurdle for electric vehicles, or EVs, is the development of advanced battery technology to extend driving range, safety and reliability.

ORNL scientists found that a small tweak created big performance improvements in a type of solid-state battery, a technology considered vital to broader electric vehicle adoption.
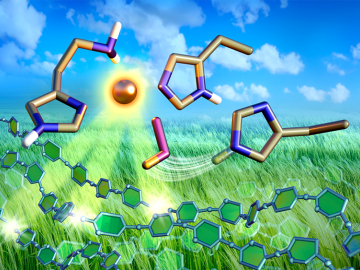
Nonfood, plant-based biofuels have potential as a green alternative to fossil fuels, but the enzymes required for production are too inefficient and costly to produce. However, new research is shining a light on enzymes from fungi that could make biofuels economically viable.

Andrew Ullman, Distinguished Staff Fellow at Oak Ridge National Laboratory, is using chemistry to devise a better battery

ORNL researchers have identified a mechanism in a 3D-printed alloy – termed “load shuffling” — that could enable the design of better-performing lightweight materials for vehicles.

The presence of minerals called ash in plants makes little difference to the fitness of new naturally derived compound materials designed for additive manufacturing, an Oak Ridge National Laboratory-led team found.

Oak Ridge National Laboratory scientists designed a recyclable polymer for carbon-fiber composites to enable circular manufacturing of parts that boost energy efficiency in automotive, wind power and aerospace applications.
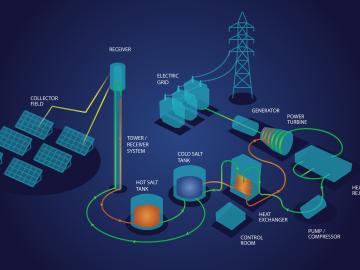
Oak Ridge National Laboratory scientists recently demonstrated a low-temperature, safe route to purifying molten chloride salts that minimizes their ability to corrode metals. This method could make the salts useful for storing energy generated from the sun’s heat.
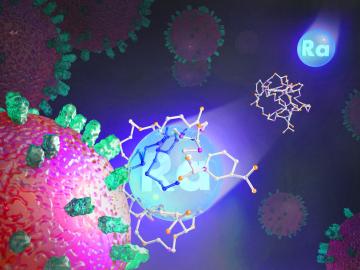
Researchers at ORNL explored radium’s chemistry to advance cancer treatments using ionizing radiation.




