Filter News
Area of Research
- (-) Materials (18)
- (-) National Security (1)
- (-) Nuclear Science and Technology (9)
- Advanced Manufacturing (1)
- Biology and Environment (7)
- Clean Energy (35)
- Computational Biology (1)
- Computer Science (1)
- Energy Sciences (1)
- Fusion and Fission (7)
- Fusion Energy (8)
- Isotopes (4)
- Materials for Computing (2)
- Neutron Science (28)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Supercomputing (7)
News Type
News Topics
- (-) Advanced Reactors (6)
- (-) Biomedical (3)
- (-) Energy Storage (9)
- (-) Fusion (5)
- (-) Neutron Science (8)
- 3-D Printing/Advanced Manufacturing (7)
- Artificial Intelligence (2)
- Big Data (2)
- Bioenergy (2)
- Buildings (1)
- Chemical Sciences (6)
- Clean Water (1)
- Climate Change (1)
- Composites (4)
- Computer Science (5)
- Coronavirus (2)
- Critical Materials (5)
- Cybersecurity (5)
- Decarbonization (1)
- Environment (3)
- Grid (3)
- Isotopes (3)
- Machine Learning (2)
- Materials (15)
- Materials Science (25)
- Microscopy (9)
- Molten Salt (4)
- Nanotechnology (12)
- National Security (10)
- Nuclear Energy (19)
- Physics (9)
- Polymers (8)
- Quantum Computing (2)
- Quantum Science (2)
- Security (3)
- Space Exploration (3)
- Summit (1)
- Sustainable Energy (4)
- Transportation (7)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.

ORNL scientists found that a small tweak created big performance improvements in a type of solid-state battery, a technology considered vital to broader electric vehicle adoption.

Andrew Ullman, Distinguished Staff Fellow at Oak Ridge National Laboratory, is using chemistry to devise a better battery

Warming a crystal of the mineral fresnoite, ORNL scientists discovered that excitations called phasons carried heat three times farther and faster than phonons, the excitations that usually carry heat through a material.
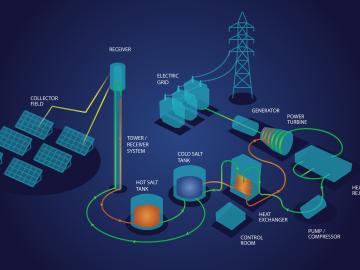
Oak Ridge National Laboratory scientists recently demonstrated a low-temperature, safe route to purifying molten chloride salts that minimizes their ability to corrode metals. This method could make the salts useful for storing energy generated from the sun’s heat.
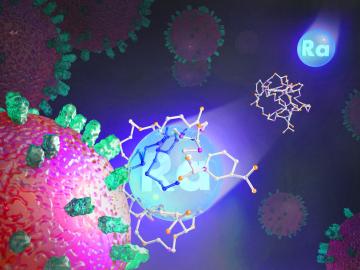
Researchers at ORNL explored radium’s chemistry to advance cancer treatments using ionizing radiation.

Marcel Demarteau is director of the Physics Division at the Department of Energy’s Oak Ridge National Laboratory. For topics from nuclear structure to astrophysics, he shapes ORNL’s physics research agenda.

A developing method to gauge the occurrence of a nuclear reactor anomaly has the potential to save millions of dollars.
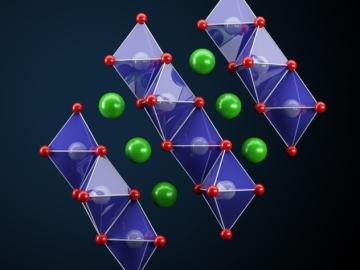
Oak Ridge National Laboratory scientists seeking the source of charge loss in lithium-ion batteries demonstrated that coupling a thin-film cathode with a solid electrolyte is a rapid way to determine the root cause.

Oak Ridge National Laboratory researchers have discovered a better way to separate actinium-227, a rare isotope essential for an FDA-approved cancer treatment.




