Filter News
Area of Research
- Advanced Manufacturing (2)
- Biology and Environment (18)
- Clean Energy (62)
- Computational Biology (1)
- Computational Engineering (2)
- Energy Frontier Research Centers (1)
- Fuel Cycle Science and Technology (1)
- Fusion and Fission (12)
- Fusion Energy (8)
- Isotope Development and Production (1)
- Isotopes (3)
- Materials (66)
- Materials for Computing (10)
- Mathematics (1)
- National Security (7)
- Neutron Science (65)
- Nuclear Science and Technology (22)
- Nuclear Systems Modeling, Simulation and Validation (2)
- Supercomputing (23)
- Transportation Systems (2)
News Type
News Topics
- (-) Advanced Reactors (23)
- (-) Biomedical (28)
- (-) Element Discovery (1)
- (-) Mathematics (1)
- (-) Molten Salt (7)
- (-) Nanotechnology (38)
- (-) Neutron Science (76)
- (-) Nuclear Energy (44)
- (-) Transportation (59)
- 3-D Printing/Advanced Manufacturing (75)
- Artificial Intelligence (42)
- Big Data (24)
- Bioenergy (39)
- Biology (39)
- Biotechnology (10)
- Buildings (32)
- Chemical Sciences (38)
- Clean Water (14)
- Climate Change (44)
- Composites (18)
- Computer Science (96)
- Coronavirus (28)
- Critical Materials (23)
- Cybersecurity (20)
- Decarbonization (26)
- Education (3)
- Energy Storage (72)
- Environment (79)
- Exascale Computing (10)
- Fossil Energy (1)
- Frontier (15)
- Fusion (23)
- Grid (35)
- High-Performance Computing (37)
- Hydropower (6)
- Irradiation (2)
- Isotopes (22)
- ITER (5)
- Machine Learning (23)
- Materials (94)
- Materials Science (83)
- Mercury (5)
- Microscopy (27)
- National Security (21)
- Net Zero (4)
- Partnerships (28)
- Physics (28)
- Polymers (21)
- Quantum Computing (13)
- Quantum Science (36)
- Renewable Energy (1)
- Security (12)
- Simulation (15)
- Space Exploration (13)
- Statistics (3)
- Summit (26)
- Sustainable Energy (75)
- Transformational Challenge Reactor (4)
Media Contacts
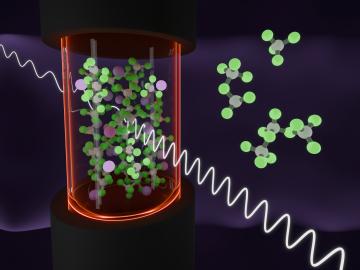
An Oak Ridge National Laboratory team revealed how chemical species form in a highly reactive molten salt mixture of aluminum chloride and potassium chloride by unraveling vibrational signatures and observing ion exchanges.

An international team using neutrons set the first benchmark (one nanosecond) for a polymer-electrolyte and lithium-salt mixture. Findings could produce safer, more powerful lithium batteries.

Jens Dilling has been named associate laboratory director for the Neutron Sciences Directorate at the Department of Energy’s Oak Ridge National Laboratory, effective April 1.

The Department of Energy’s Oak Ridge National Laboratory is providing national leadership in a new collaboration among five national laboratories to accelerate U.S. production of clean hydrogen fuel cells and electrolyzers.
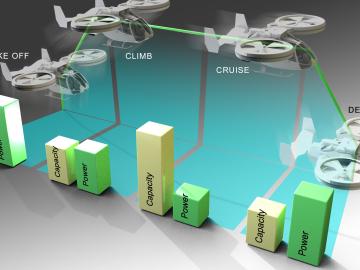
Researchers at ORNL are taking cleaner transportation to the skies by creating and evaluating new batteries for airborne electric vehicles that take off and land vertically.

A team of researchers at ORNL demonstrated that a light-duty passenger electric vehicle can be wirelessly charged at 100-kW with 96% efficiency using polyphase electromagnetic coupling coils with rotating magnetic fields.

Two different teams that included Oak Ridge National Laboratory employees were honored Feb. 20 with Secretary’s Honor Achievement Awards from the Department of Energy. This is DOE's highest form of employee recognition.

Corning uses neutron scattering to study the stability of different types of glass. Recently, researchers for the company have found that understanding the stability of the rings of atoms in glass materials can help predict the performance of glass products.

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.
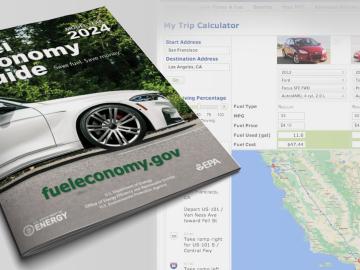
Oak Ridge National Laboratory researchers have identified the most energy-efficient 2024 model year vehicles available in the United States, including electric and hybrids, in the latest edition of the Department of Energy’s Fuel Economy Guide.




