Filter News
Area of Research
- (-) Materials (42)
- Advanced Manufacturing (3)
- Biology and Environment (18)
- Clean Energy (56)
- Computational Engineering (1)
- Computer Science (3)
- Fusion and Fission (4)
- Isotopes (1)
- Materials for Computing (7)
- National Security (11)
- Neutron Science (11)
- Nuclear Science and Technology (3)
- Quantum information Science (1)
- Supercomputing (49)
News Topics
- (-) Computer Science (8)
- (-) Energy Storage (19)
- (-) Exascale Computing (1)
- (-) Frontier (2)
- (-) High-Performance Computing (2)
- (-) Machine Learning (2)
- (-) Microscopy (12)
- (-) Polymers (6)
- (-) Sustainable Energy (7)
- (-) Transformational Challenge Reactor (1)
- 3-D Printing/Advanced Manufacturing (13)
- Advanced Reactors (1)
- Artificial Intelligence (4)
- Bioenergy (8)
- Biology (4)
- Biomedical (3)
- Buildings (2)
- Chemical Sciences (20)
- Climate Change (5)
- Composites (3)
- Coronavirus (2)
- Critical Materials (8)
- Cybersecurity (3)
- Decarbonization (4)
- Environment (7)
- Fusion (2)
- Grid (2)
- Isotopes (5)
- ITER (1)
- Materials (38)
- Materials Science (35)
- Molten Salt (2)
- Nanotechnology (21)
- National Security (3)
- Net Zero (1)
- Neutron Science (18)
- Nuclear Energy (2)
- Partnerships (8)
- Physics (14)
- Quantum Computing (1)
- Quantum Science (10)
- Renewable Energy (1)
- Security (1)
- Summit (1)
- Transportation (4)
Media Contacts
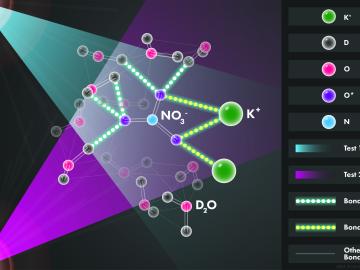
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

A scientific team led by the Department of Energy’s Oak Ridge National Laboratory has found a new way to take the local temperature of a material from an area about a billionth of a meter wide, or approximately 100,000 times thinner than a human hair. This discove...

