Filter News
Area of Research
- (-) Biology and Environment (10)
- (-) Materials (38)
- (-) National Security (12)
- Advanced Manufacturing (3)
- Clean Energy (58)
- Computational Engineering (1)
- Computer Science (7)
- Energy Sciences (1)
- Fuel Cycle Science and Technology (1)
- Fusion and Fission (12)
- Fusion Energy (6)
- Isotope Development and Production (1)
- Isotopes (11)
- Materials for Computing (4)
- Neutron Science (9)
- Nuclear Science and Technology (19)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Sensors and Controls (1)
- Supercomputing (22)
News Type
News Topics
- (-) Artificial Intelligence (12)
- (-) Energy Storage (28)
- (-) Isotopes (7)
- (-) Machine Learning (8)
- (-) Nuclear Energy (7)
- (-) Security (6)
- 3-D Printing/Advanced Manufacturing (23)
- Advanced Reactors (3)
- Big Data (4)
- Bioenergy (24)
- Biology (31)
- Biomedical (10)
- Biotechnology (7)
- Buildings (3)
- Chemical Sciences (24)
- Clean Water (4)
- Climate Change (19)
- Composites (8)
- Computer Science (24)
- Coronavirus (10)
- Critical Materials (13)
- Cybersecurity (12)
- Decarbonization (8)
- Environment (35)
- Exascale Computing (1)
- Frontier (2)
- Fusion (6)
- Grid (8)
- High-Performance Computing (11)
- Hydropower (3)
- ITER (1)
- Materials (54)
- Materials Science (56)
- Mercury (1)
- Microscopy (18)
- Molten Salt (3)
- Nanotechnology (30)
- National Security (11)
- Net Zero (2)
- Neutron Science (25)
- Partnerships (12)
- Physics (16)
- Polymers (12)
- Quantum Computing (2)
- Quantum Science (11)
- Renewable Energy (1)
- Simulation (5)
- Space Exploration (1)
- Summit (5)
- Sustainable Energy (24)
- Transformational Challenge Reactor (1)
- Transportation (13)
Media Contacts
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.
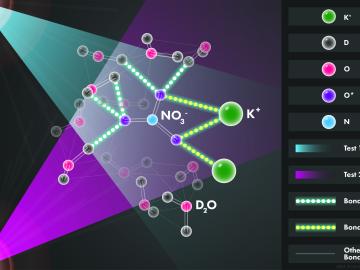
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.

A shield assembly that protects an instrument measuring ion and electron fluxes for a NASA mission to touch the Sun was tested in extreme experimental environments at Oak Ridge National Laboratory—and passed with flying colors. Components aboard Parker Solar Probe, which will endure th...




