Filter News
Area of Research
- (-) Materials (66)
- Advanced Manufacturing (1)
- Biology and Environment (18)
- Clean Energy (33)
- Computer Science (3)
- Energy Frontier Research Centers (1)
- Fusion and Fission (5)
- Isotope Development and Production (1)
- Isotopes (7)
- Materials Characterization (1)
- Materials for Computing (8)
- Materials Under Extremes (1)
- National Security (14)
- Neutron Science (46)
- Nuclear Science and Technology (4)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (41)
News Topics
- (-) Climate Change (5)
- (-) Composites (3)
- (-) Cybersecurity (3)
- (-) Frontier (2)
- (-) Isotopes (5)
- (-) Materials Science (35)
- (-) Molten Salt (2)
- (-) Nanotechnology (21)
- (-) Neutron Science (18)
- (-) Quantum Science (10)
- (-) Security (1)
- 3-D Printing/Advanced Manufacturing (13)
- Advanced Reactors (1)
- Artificial Intelligence (4)
- Bioenergy (8)
- Biology (4)
- Biomedical (4)
- Buildings (2)
- Chemical Sciences (20)
- Computer Science (9)
- Coronavirus (2)
- Critical Materials (8)
- Decarbonization (4)
- Energy Storage (19)
- Environment (7)
- Exascale Computing (1)
- Fusion (2)
- Grid (2)
- High-Performance Computing (3)
- ITER (1)
- Machine Learning (2)
- Materials (38)
- Microscopy (12)
- National Security (3)
- Net Zero (1)
- Nuclear Energy (2)
- Partnerships (8)
- Physics (14)
- Polymers (6)
- Quantum Computing (1)
- Renewable Energy (1)
- Summit (2)
- Sustainable Energy (7)
- Transformational Challenge Reactor (1)
- Transportation (4)
Media Contacts

Scientists at the Department of Energy’s Oak Ridge National Laboratory have created a recipe for a renewable 3D printing feedstock that could spur a profitable new use for an intractable biorefinery byproduct: lignin.
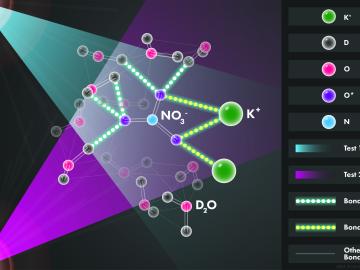
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

Scientists at the Department of Energy’s Oak Ridge National Laboratory induced a two-dimensional material to cannibalize itself for atomic “building blocks” from which stable structures formed. The findings, reported in Nature Communications, provide insights that ...

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.

A scientific team led by the Department of Energy’s Oak Ridge National Laboratory has found a new way to take the local temperature of a material from an area about a billionth of a meter wide, or approximately 100,000 times thinner than a human hair. This discove...
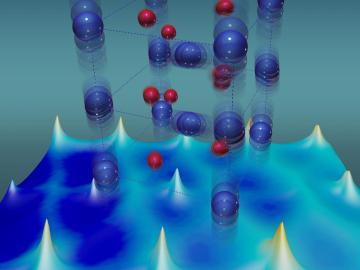
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.




