Filter News
Area of Research
- (-) Materials (56)
- Biology and Environment (12)
- Clean Energy (27)
- Computational Engineering (1)
- Computer Science (1)
- Fusion and Fission (7)
- Fusion Energy (1)
- Isotope Development and Production (1)
- Isotopes (2)
- Materials Characterization (1)
- Materials for Computing (6)
- Materials Under Extremes (1)
- National Security (5)
- Neutron Science (43)
- Nuclear Science and Technology (5)
- Supercomputing (29)
News Topics
- (-) Advanced Reactors (1)
- (-) Biomedical (3)
- (-) Critical Materials (8)
- (-) Exascale Computing (1)
- (-) Frontier (2)
- (-) Materials Science (35)
- (-) Neutron Science (18)
- 3-D Printing/Advanced Manufacturing (13)
- Artificial Intelligence (4)
- Bioenergy (8)
- Biology (4)
- Buildings (2)
- Chemical Sciences (20)
- Climate Change (5)
- Composites (3)
- Computer Science (8)
- Coronavirus (2)
- Cybersecurity (3)
- Decarbonization (4)
- Energy Storage (19)
- Environment (7)
- Fusion (2)
- Grid (2)
- High-Performance Computing (2)
- Isotopes (5)
- ITER (1)
- Machine Learning (2)
- Materials (38)
- Microscopy (12)
- Molten Salt (2)
- Nanotechnology (21)
- National Security (3)
- Net Zero (1)
- Nuclear Energy (2)
- Partnerships (8)
- Physics (14)
- Polymers (6)
- Quantum Computing (1)
- Quantum Science (10)
- Renewable Energy (1)
- Security (1)
- Summit (1)
- Sustainable Energy (7)
- Transformational Challenge Reactor (1)
- Transportation (4)
Media Contacts
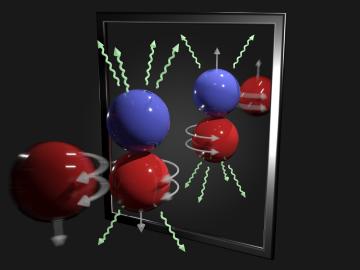
A team of scientists has for the first time measured the elusive weak interaction between protons and neutrons in the nucleus of an atom. They had chosen the simplest nucleus consisting of one neutron and one proton for the study.
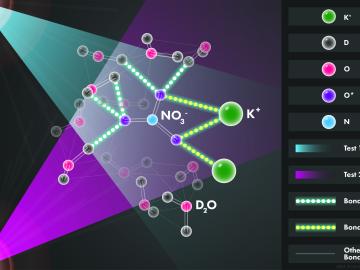
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.

Researchers at the Department of Energy’s Oak Ridge National Laboratory have demonstrated that permanent magnets produced by additive manufacturing can outperform bonded magnets made using traditional techniques while conserving critical materials. Scientists fabric...

A new technology developed by the U.S. Department of Energy’s Critical Materials Institute that aids in the recycling, recovery and extraction of rare earth minerals has been licensed to U.S. Rare Earths, Inc.
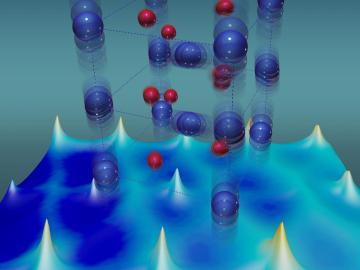
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.




