Filter News
Area of Research
- Advanced Manufacturing (3)
- Biology and Environment (11)
- Clean Energy (18)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (1)
- Fuel Cycle Science and Technology (1)
- Fusion and Fission (9)
- Fusion Energy (6)
- Isotope Development and Production (1)
- Isotopes (9)
- Materials (17)
- Materials for Computing (1)
- Mathematics (1)
- National Security (12)
- Neutron Science (3)
- Nuclear Science and Technology (20)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Quantum information Science (2)
- Renewable Energy (1)
- Supercomputing (9)
News Type
News Topics
- (-) Clean Water (14)
- (-) Cybersecurity (20)
- (-) Hydropower (6)
- (-) Isotopes (22)
- (-) Mercury (5)
- (-) Molten Salt (7)
- (-) Nuclear Energy (44)
- 3-D Printing/Advanced Manufacturing (75)
- Advanced Reactors (23)
- Artificial Intelligence (42)
- Big Data (24)
- Bioenergy (39)
- Biology (39)
- Biomedical (28)
- Biotechnology (10)
- Buildings (32)
- Chemical Sciences (38)
- Climate Change (44)
- Composites (18)
- Computer Science (96)
- Coronavirus (28)
- Critical Materials (23)
- Decarbonization (26)
- Education (3)
- Element Discovery (1)
- Energy Storage (72)
- Environment (79)
- Exascale Computing (10)
- Fossil Energy (1)
- Frontier (15)
- Fusion (23)
- Grid (35)
- High-Performance Computing (37)
- Irradiation (2)
- ITER (5)
- Machine Learning (23)
- Materials (94)
- Materials Science (83)
- Mathematics (1)
- Microscopy (27)
- Nanotechnology (38)
- National Security (21)
- Net Zero (4)
- Neutron Science (76)
- Partnerships (28)
- Physics (28)
- Polymers (21)
- Quantum Computing (13)
- Quantum Science (36)
- Renewable Energy (1)
- Security (12)
- Simulation (15)
- Space Exploration (13)
- Statistics (3)
- Summit (26)
- Sustainable Energy (75)
- Transformational Challenge Reactor (4)
- Transportation (59)
Media Contacts
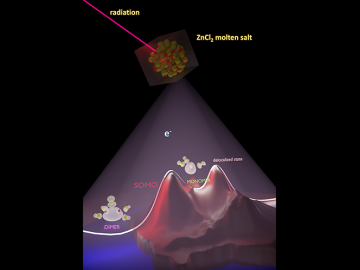
An Oak Ridge National Laboratory team revealed how chemical species form in a highly reactive molten salt mixture of aluminum chloride and potassium chloride by unraveling vibrational signatures and observing ion exchanges.

Scientists have uncovered the properties of a rare earth element that was first discovered 80 years ago at the very same laboratory, opening a new pathway for the exploration of elements critical in modern technology, from medicine to space travel.
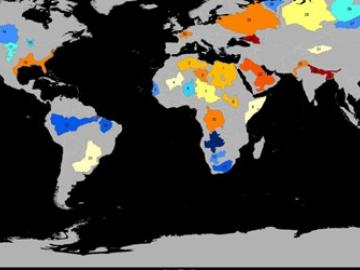
Groundwater withdrawals are expected to peak in about one-third of the world’s basins by 2050, potentially triggering significant trade and agriculture shifts, a new analysis finds.

Two different teams that included Oak Ridge National Laboratory employees were honored Feb. 20 with Secretary’s Honor Achievement Awards from the Department of Energy. This is DOE's highest form of employee recognition.
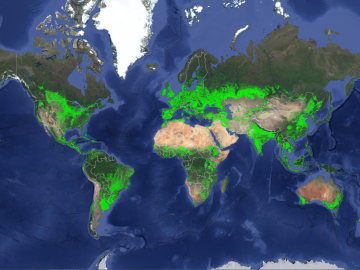
ORNL climate modeling expertise contributed to a project that assessed global emissions of ammonia from croplands now and in a warmer future, while also identifying solutions tuned to local growing conditions.

As vehicles gain technological capabilities, car manufacturers are using an increasing number of computers and sensors to improve situational awareness and enhance the driving experience.
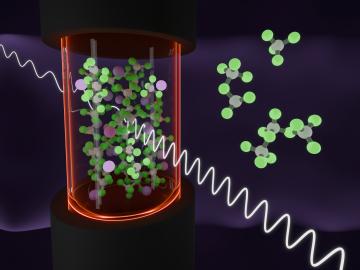
In a finding that helps elucidate how molten salts in advanced nuclear reactors might behave, scientists have shown how electrons interacting with the ions of the molten salt can form three states with different properties. Understanding these states can help predict the impact of radiation on the performance of salt-fueled reactors.
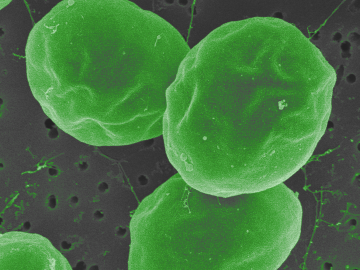
In the search for ways to fight methylmercury in global waterways, scientists at Oak Ridge National Laboratory discovered that some forms of phytoplankton are good at degrading the potent neurotoxin.

Like most scientists, Chengping Chai is not content with the surface of things: He wants to probe beyond to learn what’s really going on. But in his case, he is literally building a map of the world beneath, using seismic and acoustic data that reveal when and where the earth moves.

ORNL’s electromagnetic isotope separator, or EMIS, made history in 2018 when it produced 500 milligrams of the rare isotope ruthenium-96, unavailable anywhere else in the world.




