Filter News
Area of Research
- Biology and Environment (40)
- Clean Energy (38)
- Climate and Environmental Systems (4)
- Computational Biology (1)
- Computational Engineering (2)
- Computer Science (1)
- Fusion and Fission (1)
- Isotopes (3)
- Materials (38)
- Materials for Computing (5)
- Mathematics (1)
- National Security (6)
- Neutron Science (63)
- Nuclear Science and Technology (7)
- Supercomputing (30)
News Type
News Topics
- (-) Biomedical (28)
- (-) Environment (79)
- (-) Exascale Computing (10)
- (-) Molten Salt (7)
- (-) Neutron Science (76)
- 3-D Printing/Advanced Manufacturing (75)
- Advanced Reactors (23)
- Artificial Intelligence (42)
- Big Data (24)
- Bioenergy (39)
- Biology (39)
- Biotechnology (10)
- Buildings (32)
- Chemical Sciences (38)
- Clean Water (14)
- Climate Change (44)
- Composites (18)
- Computer Science (96)
- Coronavirus (28)
- Critical Materials (23)
- Cybersecurity (20)
- Decarbonization (27)
- Education (3)
- Element Discovery (1)
- Energy Storage (72)
- Fossil Energy (1)
- Frontier (15)
- Fusion (23)
- Grid (35)
- High-Performance Computing (37)
- Hydropower (6)
- Irradiation (2)
- Isotopes (22)
- ITER (5)
- Machine Learning (23)
- Materials (94)
- Materials Science (83)
- Mathematics (1)
- Mercury (5)
- Microscopy (27)
- Nanotechnology (38)
- National Security (21)
- Net Zero (4)
- Nuclear Energy (44)
- Partnerships (28)
- Physics (28)
- Polymers (21)
- Quantum Computing (13)
- Quantum Science (36)
- Renewable Energy (1)
- Security (12)
- Simulation (15)
- Space Exploration (13)
- Statistics (3)
- Summit (26)
- Sustainable Energy (75)
- Transformational Challenge Reactor (4)
- Transportation (60)
Media Contacts
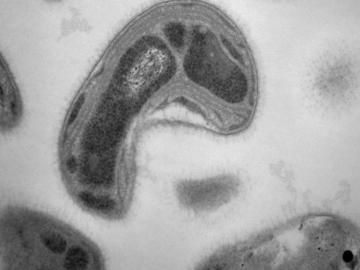
A team led by the Department of Energy’s Oak Ridge National Laboratory has identified a novel microbial process that can break down toxic methylmercury in the environment, a fundamental scientific discovery that could potentially reduce mercury toxicity levels and sup...

The Department of Energy’s Oak Ridge National Laboratory has received funding from DOE’s Exascale Computing Project (ECP) to develop applications for future exascale systems that will be 50 to 100 times more powerful than today’s fastest supercomputers.

An Oak Ridge National Laboratory study is providing an unprecedented watershed-scale understanding of mercury in soils and sediments. Researchers focused on evaluating mercury and soil properties along the banks of a mercury-contaminated stream in Oak Ridge, Tenn., sampling 145 loca...

Environmental scientists can more efficiently detect genes required to convert mercury in the environment into more toxic methylmercury with molecular probes developed by researchers at the Department of Energy’s Oak Ridge National Laboratory. “We now have a quic...
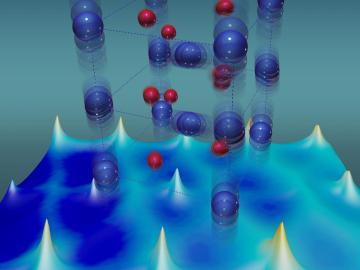
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.

