Filter News
Area of Research
- (-) Materials (19)
- (-) Neutron Science (9)
- Advanced Manufacturing (2)
- Biology and Environment (45)
- Clean Energy (38)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (2)
- Electricity and Smart Grid (1)
- Functional Materials for Energy (1)
- Fusion and Fission (2)
- Isotope Development and Production (1)
- Isotopes (15)
- Materials for Computing (4)
- National Security (23)
- Nuclear Science and Technology (5)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (32)
News Type
News Topics
- (-) Cybersecurity (4)
- (-) Environment (13)
- (-) Isotopes (5)
- (-) Machine Learning (3)
- (-) Security (2)
- (-) Space Exploration (1)
- (-) Summit (4)
- 3-D Printing/Advanced Manufacturing (15)
- Advanced Reactors (3)
- Artificial Intelligence (5)
- Big Data (1)
- Bioenergy (10)
- Biology (8)
- Biomedical (7)
- Biotechnology (1)
- Buildings (3)
- Chemical Sciences (22)
- Climate Change (5)
- Composites (3)
- Computer Science (11)
- Coronavirus (7)
- Critical Materials (8)
- Decarbonization (6)
- Energy Storage (22)
- Exascale Computing (1)
- Frontier (4)
- Fusion (4)
- Grid (3)
- High-Performance Computing (4)
- Irradiation (1)
- ITER (1)
- Materials (48)
- Materials Science (47)
- Microscopy (17)
- Molten Salt (2)
- Nanotechnology (27)
- National Security (3)
- Net Zero (1)
- Neutron Science (53)
- Nuclear Energy (6)
- Partnerships (9)
- Physics (22)
- Polymers (8)
- Quantum Computing (2)
- Quantum Science (12)
- Renewable Energy (1)
- Simulation (1)
- Sustainable Energy (9)
- Transformational Challenge Reactor (1)
- Transportation (8)
Media Contacts
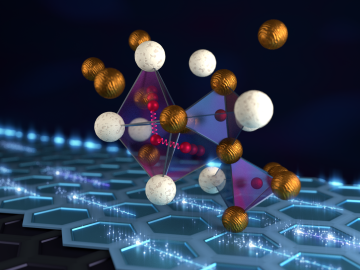
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.
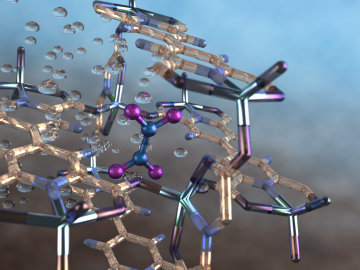
An international team of scientists, led by the University of Manchester, has developed a metal-organic framework, or MOF, material

Researchers at the Department of Energy’s Oak Ridge National Laboratory have received five 2019 R&D 100 Awards, increasing the lab’s total to 221 since the award’s inception in 1963.
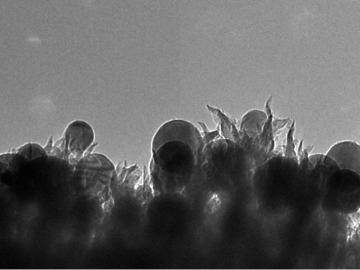
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
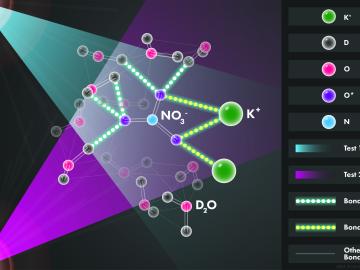
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.




