Filter News
Area of Research
- (-) Climate and Environmental Systems (2)
- (-) Energy Frontier Research Centers (7)
- Advanced Manufacturing (14)
- Biological Systems (15)
- Biology and Environment (42)
- Biology and Soft Matter (1)
- Building Technologies (3)
- Chemistry and Physics at Interfaces (4)
- Clean Energy (187)
- Computational Biology (4)
- Computational Engineering (2)
- Computer Science (6)
- Electricity and Smart Grid (1)
- Energy Sciences (3)
- Fossil Energy (2)
- Fuel Cycle Science and Technology (1)
- Functional Materials for Energy (8)
- Fusion and Fission (19)
- Fusion Energy (2)
- Geographic Information Science and Technology (2)
- Isotope Development and Production (1)
- Isotopes (10)
- Materials (226)
- Materials Characterization (2)
- Materials for Computing (13)
- Materials Synthesis from Atoms to Systems (5)
- Materials Under Extremes (5)
- National Security (25)
- Neutron Science (80)
- Nuclear Science and Technology (27)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Nuclear Systems Technology (1)
- Quantum Condensed Matter (1)
- Quantum information Science (2)
- Reactor Technology (1)
- Sensors and Controls (1)
- Supercomputing (115)
- Transportation Systems (5)
News Topics
Media Contacts

Scientists at the Department of Energy’s Oak Ridge National Laboratory induced a two-dimensional material to cannibalize itself for atomic “building blocks” from which stable structures formed. The findings, reported in Nature Communications, provide insights that ...

A futuristic experiment simulating warmer environmental conditions has shown that peatland vegetation responds to higher temperatures with an earlier and longer growth period.
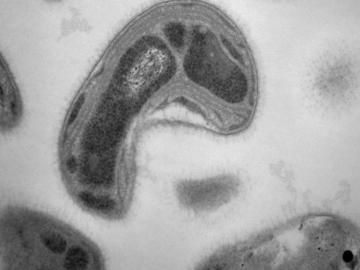
A team led by the Department of Energy’s Oak Ridge National Laboratory has identified a novel microbial process that can break down toxic methylmercury in the environment, a fundamental scientific discovery that could potentially reduce mercury toxicity levels and sup...


Less than 1 percent of Earth’s water is drinkable. Removing salt and other minerals from our biggest available source of water—seawater—may help satisfy a growing global population thirsty for fresh water for drinking, farming, transportation, heating, cooling and industry. But desalination is an energy-intensive process, which concerns those wanting to expand its application.

Graphene, a strong, lightweight carbon honeycombed structure that’s only one atom thick, holds great promise for energy research and development. Recently scientists with the Fluid Interface Reactions, Structures, and Transport (FIRST) Energy Frontier Research Center (EFRC), led by the US Department of Energy’s Oak Ridge National Laboratory, revealed graphene can serve as a proton-selective permeable membrane, providing a new basis for streamlined and more efficient energy technologies such as improved fuel cells.

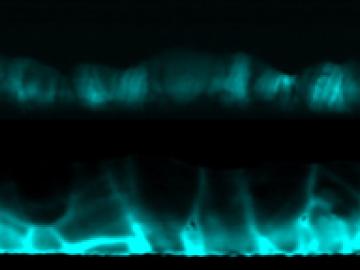
Treating cadmium-telluride (CdTe) solar cell materials with cadmium-chloride improves their efficiency, but researchers have not fully understood why.

Using a new microscopy method, researchers at the Department of Energy’s Oak Ridge National Laboratory can image and measure electrochemical processes in batteries in real time and at nanoscale resolution.




