Filter News
Area of Research
- (-) Fusion Energy (1)
- (-) Materials (44)
- (-) National Security (55)
- Advanced Manufacturing (4)
- Biology and Environment (28)
- Clean Energy (41)
- Computational Biology (1)
- Computational Engineering (1)
- Computer Science (5)
- Fuel Cycle Science and Technology (1)
- Fusion and Fission (9)
- Isotopes (2)
- Materials for Computing (10)
- Neutron Science (80)
- Nuclear Science and Technology (9)
- Quantum information Science (2)
- Sensors and Controls (1)
- Supercomputing (97)
News Type
News Topics
- (-) Advanced Reactors (4)
- (-) Artificial Intelligence (20)
- (-) Computer Science (30)
- (-) Cybersecurity (19)
- (-) Machine Learning (15)
- (-) National Security (33)
- (-) Neutron Science (29)
- (-) Security (10)
- (-) Transformational Challenge Reactor (3)
- 3-D Printing/Advanced Manufacturing (19)
- Big Data (5)
- Bioenergy (13)
- Biology (8)
- Biomedical (6)
- Biotechnology (1)
- Buildings (4)
- Chemical Sciences (27)
- Clean Water (2)
- Climate Change (9)
- Composites (5)
- Coronavirus (4)
- Critical Materials (8)
- Decarbonization (7)
- Energy Storage (25)
- Environment (17)
- Exascale Computing (2)
- Frontier (2)
- Fusion (10)
- Grid (8)
- High-Performance Computing (7)
- Isotopes (11)
- ITER (1)
- Materials (58)
- Materials Science (52)
- Mathematics (1)
- Microscopy (18)
- Molten Salt (2)
- Nanotechnology (29)
- Net Zero (1)
- Nuclear Energy (19)
- Partnerships (14)
- Physics (25)
- Polymers (10)
- Quantum Computing (2)
- Quantum Science (11)
- Renewable Energy (1)
- Simulation (1)
- Space Exploration (1)
- Summit (3)
- Sustainable Energy (11)
- Transportation (9)
Media Contacts
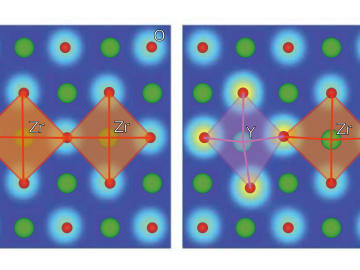
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

Scientists at the Department of Energy’s Oak Ridge National Laboratory are working to understand both the complex nature of uranium and the various oxide forms it can take during processing steps that might occur throughout the nuclear fuel cycle.
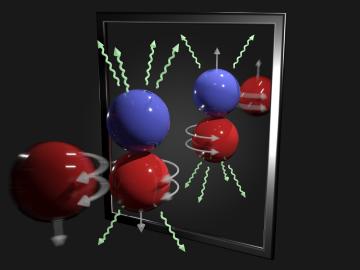
A team of scientists has for the first time measured the elusive weak interaction between protons and neutrons in the nucleus of an atom. They had chosen the simplest nucleus consisting of one neutron and one proton for the study.
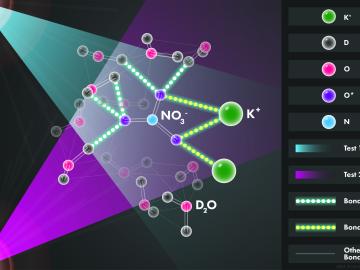
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come
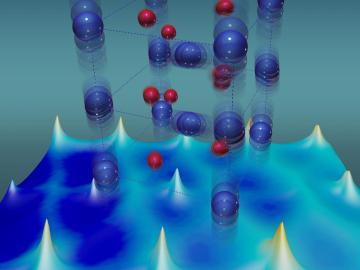
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.

