Filter News
Area of Research
- (-) Neutron Science (11)
- (-) Quantum information Science (1)
- Advanced Manufacturing (3)
- Biological Systems (1)
- Biology and Environment (29)
- Clean Energy (47)
- Electricity and Smart Grid (1)
- Fusion and Fission (4)
- Isotopes (1)
- Materials (22)
- Materials for Computing (5)
- National Security (8)
- Nuclear Science and Technology (6)
- Supercomputing (14)
News Topics
- (-) 3-D Printing/Advanced Manufacturing (3)
- (-) Bioenergy (3)
- (-) Materials Science (7)
- Artificial Intelligence (4)
- Big Data (1)
- Biology (1)
- Biomedical (5)
- Chemical Sciences (1)
- Clean Water (2)
- Computer Science (8)
- Coronavirus (3)
- Decarbonization (1)
- Energy Storage (2)
- Environment (3)
- Fossil Energy (1)
- High-Performance Computing (1)
- Machine Learning (3)
- Materials (5)
- Mathematics (1)
- Microscopy (3)
- Nanotechnology (3)
- National Security (1)
- Neutron Science (33)
- Nuclear Energy (1)
- Physics (1)
- Polymers (1)
- Quantum Computing (1)
- Quantum Science (5)
- Security (1)
- Space Exploration (1)
- Summit (2)
- Sustainable Energy (1)
- Transportation (1)
Media Contacts
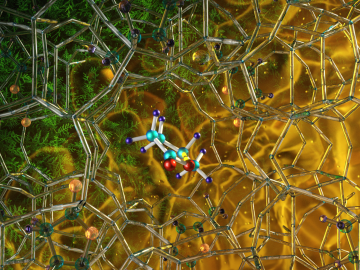
Illustration of the optimized zeolite catalyst, or NbAlS-1, which enables a highly efficient chemical reaction to create butene, a renewable source of energy, without expending high amounts of energy for the conversion. Credit: Jill Hemman, Oak Ridge National Laboratory/U.S. Dept. of Energy

Scientists at the U.S. Department of Energy’s Brookhaven National Laboratory have new experimental evidence and a predictive theory that solves a long-standing materials science mystery: why certain crystalline materials shrink when heated.

