Filter News
Area of Research
- (-) Materials (32)
- (-) Mathematics (1)
- Advanced Manufacturing (5)
- Biology and Environment (24)
- Building Technologies (2)
- Clean Energy (71)
- Climate and Environmental Systems (1)
- Computational Engineering (3)
- Computer Science (12)
- Energy Sciences (1)
- Fusion and Fission (3)
- Fusion Energy (2)
- Isotopes (10)
- Materials for Computing (7)
- National Security (11)
- Neutron Science (7)
- Nuclear Science and Technology (5)
- Quantum information Science (4)
- Supercomputing (50)
News Topics
- (-) Clean Water (2)
- (-) Computer Science (10)
- (-) Critical Materials (13)
- (-) Isotopes (7)
- (-) Sustainable Energy (10)
- 3-D Printing/Advanced Manufacturing (19)
- Advanced Reactors (2)
- Artificial Intelligence (4)
- Bioenergy (9)
- Biology (4)
- Biomedical (5)
- Buildings (3)
- Chemical Sciences (24)
- Climate Change (6)
- Composites (7)
- Coronavirus (3)
- Cybersecurity (3)
- Decarbonization (5)
- Energy Storage (26)
- Environment (9)
- Exascale Computing (1)
- Frontier (2)
- Fusion (4)
- Grid (2)
- High-Performance Computing (2)
- ITER (1)
- Machine Learning (2)
- Materials (50)
- Materials Science (54)
- Mathematics (1)
- Microscopy (18)
- Molten Salt (3)
- Nanotechnology (29)
- National Security (3)
- Net Zero (1)
- Neutron Science (22)
- Nuclear Energy (5)
- Partnerships (8)
- Physics (16)
- Polymers (12)
- Quantum Computing (2)
- Quantum Science (11)
- Renewable Energy (1)
- Security (1)
- Space Exploration (1)
- Summit (1)
- Transformational Challenge Reactor (1)
- Transportation (10)
Media Contacts

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.

Energy storage startup SPARKZ Inc. has exclusively licensed five battery technologies from the Department of Energy’s Oak Ridge National Laboratory designed to eliminate cobalt metal in lithium-ion batteries. The advancement is aimed at accelerating the production of electric vehicles and energy storage solutions for the power grid.
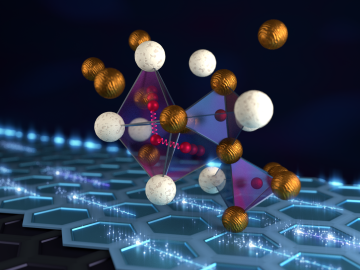
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.

Researchers at the Department of Energy’s Oak Ridge National Laboratory have received five 2019 R&D 100 Awards, increasing the lab’s total to 221 since the award’s inception in 1963.

Quanex Building Products has signed a non-exclusive agreement to license a method to produce insulating material from ORNL. The low-cost material can be used as an additive to increase thermal insulation performance and improve energy efficiency when applied to a variety of building products.
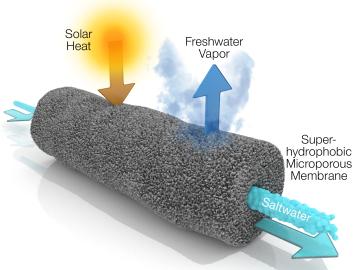
A new method developed at Oak Ridge National Laboratory improves the energy efficiency of a desalination process known as solar-thermal evaporation.

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
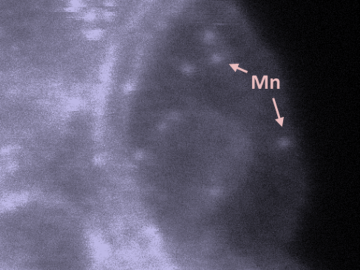
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.
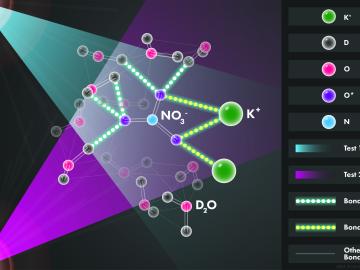
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come




