Filter News
Area of Research
- (-) Neutron Science (5)
- Advanced Manufacturing (1)
- Biology and Environment (6)
- Clean Energy (24)
- Computational Engineering (1)
- Computer Science (1)
- Fusion and Fission (2)
- Isotopes (4)
- Materials (7)
- Materials for Computing (3)
- National Security (4)
- Nuclear Science and Technology (6)
- Supercomputing (9)
News Topics
- (-) Biomedical (3)
- (-) Environment (2)
- 3-D Printing/Advanced Manufacturing (1)
- Advanced Reactors (1)
- Bioenergy (2)
- Biology (1)
- Climate Change (1)
- Computer Science (5)
- Coronavirus (4)
- Fusion (1)
- Materials (1)
- Materials Science (9)
- Microscopy (1)
- Nanotechnology (6)
- National Security (1)
- Neutron Science (21)
- Physics (3)
- Quantum Science (4)
- Summit (4)
- Sustainable Energy (1)
- Transportation (1)
Media Contacts
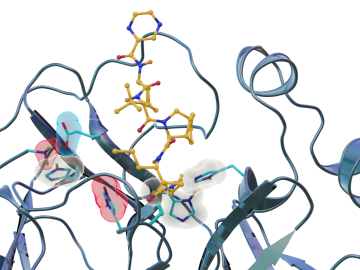
Scientists have found new, unexpected behaviors when SARS-CoV-2 – the virus that causes COVID-19 – encounters drugs known as inhibitors, which bind to certain components of the virus and block its ability to reproduce.
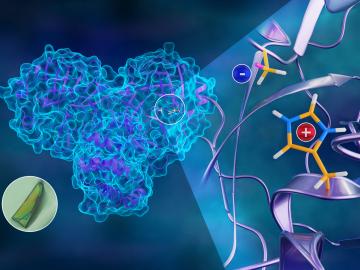
To better understand how the novel coronavirus behaves and how it can be stopped, scientists have completed a three-dimensional map that reveals the location of every atom in an enzyme molecule critical to SARS-CoV-2 reproduction.

Scientists at ORNL used neutron scattering and supercomputing to better understand how an organic solvent and water work together to break down plant biomass, creating a pathway to significantly improve the production of renewable
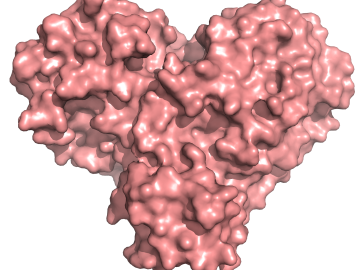
A team of researchers has performed the first room-temperature X-ray measurements on the SARS-CoV-2 main protease — the enzyme that enables the virus to reproduce.

The Department of Energy’s Office of Science has selected three Oak Ridge National Laboratory scientists for Early Career Research Program awards.




