Filter News
Area of Research
News Topics
- (-) Energy Storage (4)
- (-) Isotopes (1)
- (-) Molten Salt (1)
- 3-D Printing/Advanced Manufacturing (5)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Bioenergy (2)
- Clean Water (2)
- Composites (1)
- Computer Science (3)
- Cybersecurity (1)
- Environment (5)
- Fusion (2)
- Materials Science (15)
- Microscopy (4)
- Nanotechnology (5)
- Neutron Science (4)
- Nuclear Energy (5)
- Physics (1)
- Polymers (1)
- Quantum Science (2)
- Sustainable Energy (5)
- Transportation (5)
Media Contacts
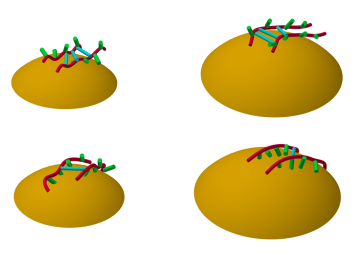
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
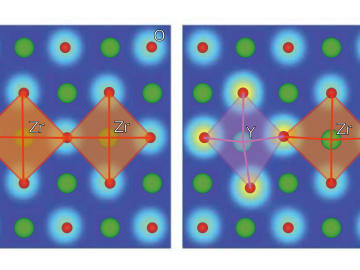
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
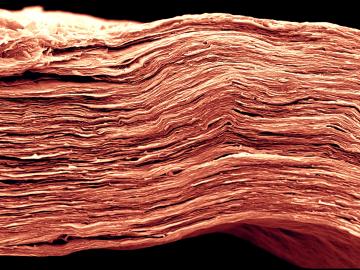

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
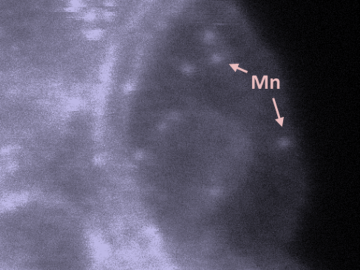
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.




