Filter News
Area of Research
News Topics
- (-) Composites (1)
- (-) Microscopy (2)
- (-) Molten Salt (1)
- (-) Polymers (1)
- 3-D Printing/Advanced Manufacturing (2)
- Advanced Reactors (1)
- Big Data (1)
- Biomedical (1)
- Clean Water (1)
- Energy Storage (4)
- Environment (1)
- Fusion (2)
- Grid (1)
- Materials Science (8)
- Nanotechnology (2)
- Neutron Science (8)
- Nuclear Energy (2)
- Physics (1)
- Quantum Science (1)
- Sustainable Energy (2)
- Transportation (4)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.
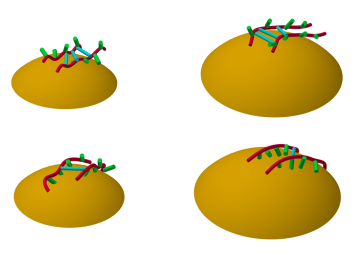
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
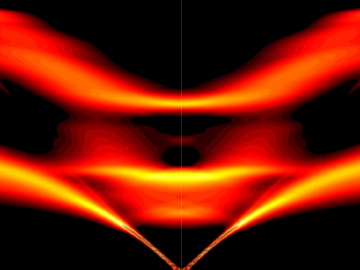
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.
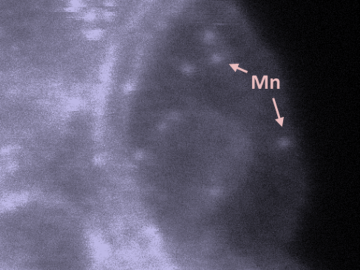
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.




