Filter News
Area of Research
- (-) Climate and Environmental Systems (1)
- (-) Materials (12)
- Biological Systems (1)
- Biology and Environment (6)
- Clean Energy (22)
- Computational Engineering (1)
- Computer Science (8)
- Fusion Energy (2)
- Isotopes (1)
- Materials for Computing (1)
- National Security (5)
- Neutron Science (8)
- Nuclear Science and Technology (3)
- Quantum information Science (4)
- Supercomputing (37)
News Topics
- (-) Bioenergy (1)
- (-) Biomedical (1)
- (-) Computer Science (7)
- (-) Molten Salt (1)
- (-) Nanotechnology (6)
- 3-D Printing/Advanced Manufacturing (8)
- Advanced Reactors (2)
- Artificial Intelligence (2)
- Big Data (2)
- Biology (1)
- Clean Water (2)
- Climate Change (1)
- Composites (1)
- Coronavirus (1)
- Cybersecurity (1)
- Energy Storage (7)
- Environment (9)
- Exascale Computing (1)
- Fusion (2)
- Machine Learning (2)
- Materials Science (23)
- Mathematics (1)
- Microscopy (5)
- Neutron Science (6)
- Nuclear Energy (8)
- Physics (5)
- Polymers (3)
- Security (1)
- Summit (1)
- Sustainable Energy (4)
- Transformational Challenge Reactor (2)
- Transportation (6)
Media Contacts
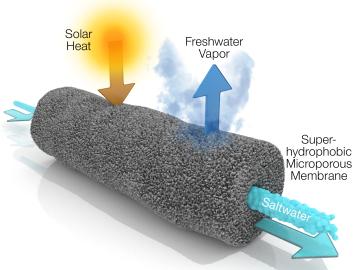
A new method developed at Oak Ridge National Laboratory improves the energy efficiency of a desalination process known as solar-thermal evaporation.
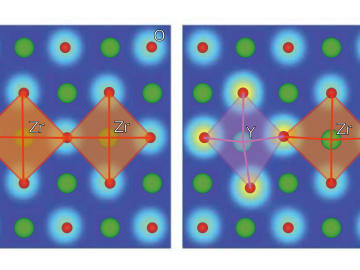
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.

