Filter News
Area of Research
News Type
News Topics
- (-) Energy Storage (4)
- (-) Environment (5)
- 3-D Printing/Advanced Manufacturing (5)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Bioenergy (2)
- Clean Water (2)
- Composites (1)
- Computer Science (3)
- Cybersecurity (1)
- Fusion (2)
- Isotopes (1)
- Materials Science (18)
- Microscopy (5)
- Molten Salt (1)
- Nanotechnology (6)
- Neutron Science (4)
- Nuclear Energy (6)
- Physics (2)
- Polymers (2)
- Quantum Science (2)
- Sustainable Energy (5)
- Transportation (5)
Media Contacts

Students often participate in internships and receive formal training in their chosen career fields during college, but some pursue professional development opportunities even earlier.
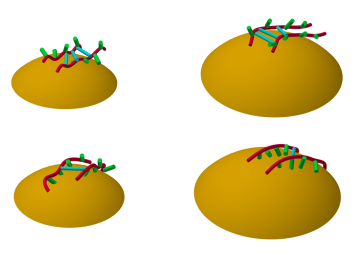
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.
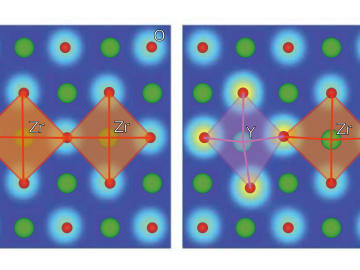
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and
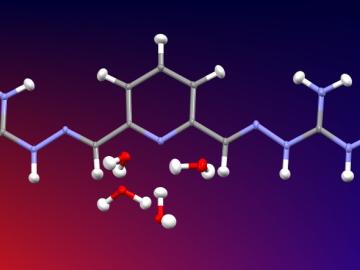
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate the effectiveness of a novel crystallization method to capture carbon dioxide directly from the air.
Scientists at the Department of Energy’s Oak Ridge National Laboratory (ORNL) have developed a process that could remove CO2 from coal-burning power plant emissions in a way that is similar to how soda lime works in scuba diving rebreathers. Their research, published January 31 in...
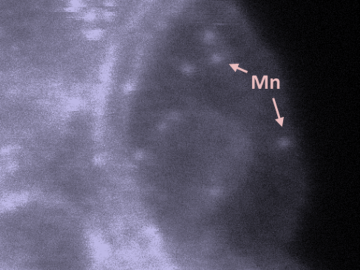
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.




