Filter News
Area of Research
- (-) Advanced Manufacturing (4)
- (-) Materials (25)
- (-) Supercomputing (8)
- (-) Transportation Systems (1)
- Biology and Environment (2)
- Clean Energy (24)
- Climate and Environmental Systems (2)
- Computer Science (2)
- Fusion Energy (2)
- National Security (1)
- Neutron Science (7)
- Quantum information Science (3)
News Topics
- (-) Environment (6)
- (-) Grid (1)
- (-) Materials Science (19)
- (-) Quantum Science (5)
- (-) Sustainable Energy (6)
- 3-D Printing/Advanced Manufacturing (8)
- Advanced Reactors (2)
- Artificial Intelligence (6)
- Big Data (4)
- Bioenergy (3)
- Biomedical (2)
- Clean Water (2)
- Composites (2)
- Computer Science (24)
- Cybersecurity (2)
- Energy Storage (5)
- Exascale Computing (2)
- Frontier (2)
- Fusion (2)
- Isotopes (1)
- Microscopy (5)
- Molten Salt (1)
- Nanotechnology (6)
- Neutron Science (5)
- Nuclear Energy (8)
- Physics (3)
- Polymers (2)
- Security (1)
- Space Exploration (2)
- Summit (9)
- Transportation (6)
Media Contacts
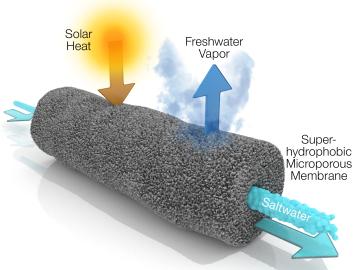
A new method developed at Oak Ridge National Laboratory improves the energy efficiency of a desalination process known as solar-thermal evaporation.
A team of scientists led by Oak Ridge National Laboratory have discovered the specific gene that controls an important symbiotic relationship between plants and soil fungi, and successfully facilitated the symbiosis in a plant that
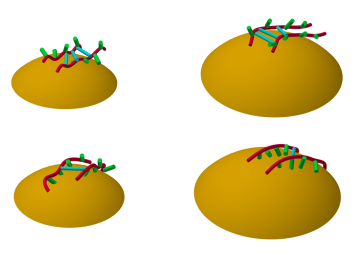
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

Oak Ridge National Laboratory has teamed with Cornell College and the University of Tennessee to study ways to repurpose waste soft drinks for carbon capture that could help cut carbon dioxide emissions.
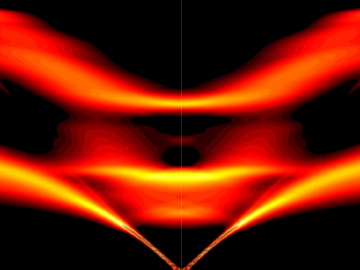
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

A team led by scientists at the Department of Energy’s Oak Ridge National Laboratory explored how atomically thin two-dimensional (2D) crystals can grow over 3D objects and how the curvature of those objects can stretch and strain the

OAK RIDGE, Tenn., May 7, 2019—Energy Secretary Rick Perry, Congressman Chuck Fleischmann and lab officials today broke ground on a multipurpose research facility that will provide state-of-the-art laboratory space

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.

Kevin Field at the Department of Energy’s Oak Ridge National Laboratory synthesizes and scrutinizes materials for nuclear power systems that must perform safely and efficiently over decades of irradiation.