Filter News
Area of Research
News Type
News Topics
- (-) Bioenergy (9)
- (-) Composites (1)
- (-) Energy Storage (4)
- (-) Environment (12)
- (-) Frontier (2)
- (-) Mercury (1)
- (-) Physics (3)
- 3-D Printing/Advanced Manufacturing (9)
- Advanced Reactors (2)
- Artificial Intelligence (7)
- Big Data (2)
- Biomedical (4)
- Biotechnology (1)
- Clean Water (3)
- Computer Science (19)
- Cybersecurity (4)
- Exascale Computing (2)
- Fusion (2)
- Grid (2)
- Isotopes (1)
- Machine Learning (2)
- Materials Science (8)
- Microscopy (1)
- Nanotechnology (3)
- Neutron Science (10)
- Nuclear Energy (6)
- Quantum Science (7)
- Security (2)
- Space Exploration (1)
- Summit (7)
- Sustainable Energy (6)
- Transportation (5)
Media Contacts

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.
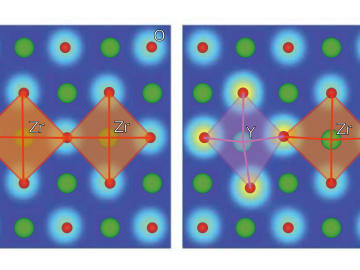
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

OAK RIDGE, Tenn., March 11, 2019—An international collaboration including scientists at the Department of Energy’s Oak Ridge National Laboratory solved a 50-year-old puzzle that explains why beta decays of atomic nuclei
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

The use of lithium-ion batteries has surged in recent years, starting with electronics and expanding into many applications, including the growing electric and hybrid vehicle industry. But the technologies to optimize recycling of these batteries have not kept pace.
Scientists at the Department of Energy’s Oak Ridge National Laboratory (ORNL) have developed a process that could remove CO2 from coal-burning power plant emissions in a way that is similar to how soda lime works in scuba diving rebreathers. Their research, published January 31 in...
![Coexpression_hi-res_image[1].jpg Coexpression_hi-res_image[1].jpg](/sites/default/files/styles/list_page_thumbnail/public/Coexpression_hi-res_image%5B1%5D_0.jpg?itok=OnLe-krT)
While studying the genes in poplar trees that control callus formation, scientists at Oak Ridge National Laboratory have uncovered genetic networks at the root of tumor formation in several human cancers.




