
Filter News
Area of Research
News Topics
- (-) Bioenergy (3)
- (-) Composites (3)
- (-) Molten Salt (2)
- (-) Nuclear Energy (10)
- 3-D Printing/Advanced Manufacturing (9)
- Advanced Reactors (2)
- Artificial Intelligence (1)
- Biomedical (1)
- Buildings (3)
- Chemical Sciences (16)
- Clean Water (2)
- Climate Change (3)
- Computer Science (4)
- Coronavirus (1)
- Critical Materials (4)
- Cybersecurity (1)
- Decarbonization (4)
- Energy Storage (9)
- Environment (8)
- Fusion (4)
- Grid (2)
- Irradiation (1)
- Isotopes (4)
- Materials (40)
- Materials Science (25)
- Microscopy (8)
- Nanotechnology (9)
- Net Zero (1)
- Neutron Science (12)
- Partnerships (6)
- Physics (11)
- Polymers (6)
- Quantum Computing (2)
- Quantum Science (3)
- Renewable Energy (1)
- Sustainable Energy (6)
- Transportation (7)
Media Contacts
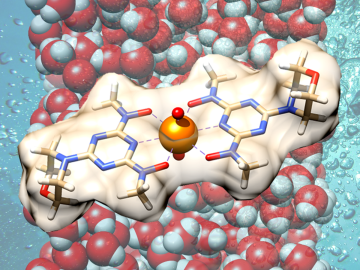
Scientists have demonstrated a new bio-inspired material for an eco-friendly and cost-effective approach to recovering uranium from seawater.

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.

Kevin Field at the Department of Energy’s Oak Ridge National Laboratory synthesizes and scrutinizes materials for nuclear power systems that must perform safely and efficiently over decades of irradiation.
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

Scientists have tested a novel heat-shielding graphite foam, originally created at Oak Ridge National Laboratory, at Germany’s Wendelstein 7-X stellarator with promising results for use in plasma-facing components of fusion reactors.

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.


