Filter News
Area of Research
- (-) Energy Sciences (1)
- (-) Isotopes (1)
- Biology and Environment (7)
- Clean Energy (72)
- Computer Science (2)
- Electricity and Smart Grid (1)
- Functional Materials for Energy (2)
- Fusion and Fission (4)
- Materials (34)
- Materials for Computing (4)
- National Security (2)
- Neutron Science (6)
- Supercomputing (8)
News Type
Media Contacts
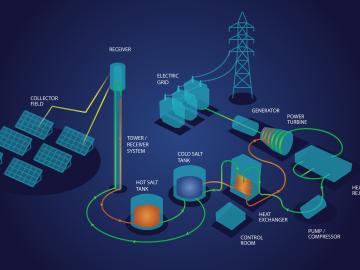
Oak Ridge National Laboratory scientists recently demonstrated a low-temperature, safe route to purifying molten chloride salts that minimizes their ability to corrode metals. This method could make the salts useful for storing energy generated from the sun’s heat.
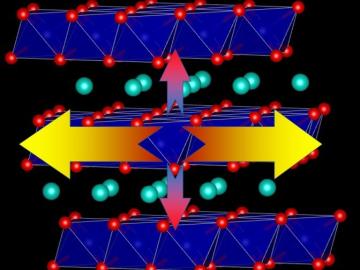
Oak Ridge National Laboratory researchers proved that the heat transport ability of lithium-ion battery cathodes is much lower than previously determined, a finding that could help explain barriers to increasing energy storage capacity and boosting performance.




