Filter News
Area of Research
News Topics
- (-) Energy Storage (41)
- (-) Neutron Science (49)
- 3-D Printing/Advanced Manufacturing (44)
- Advanced Reactors (10)
- Artificial Intelligence (29)
- Big Data (8)
- Bioenergy (24)
- Biology (22)
- Biomedical (17)
- Biotechnology (7)
- Buildings (13)
- Chemical Sciences (29)
- Clean Water (1)
- Climate Change (22)
- Composites (9)
- Computer Science (57)
- Coronavirus (17)
- Critical Materials (11)
- Cybersecurity (17)
- Decarbonization (18)
- Education (3)
- Element Discovery (1)
- Environment (36)
- Exascale Computing (9)
- Fossil Energy (1)
- Frontier (14)
- Fusion (14)
- Grid (15)
- High-Performance Computing (26)
- Isotopes (17)
- ITER (2)
- Machine Learning (13)
- Materials (59)
- Materials Science (50)
- Mercury (2)
- Microscopy (16)
- Molten Salt (2)
- Nanotechnology (26)
- National Security (18)
- Net Zero (3)
- Nuclear Energy (25)
- Partnerships (26)
- Physics (24)
- Polymers (12)
- Quantum Computing (9)
- Quantum Science (26)
- Renewable Energy (1)
- Security (11)
- Simulation (8)
- Space Exploration (3)
- Statistics (2)
- Summit (20)
- Sustainable Energy (31)
- Transformational Challenge Reactor (4)
- Transportation (24)
Media Contacts
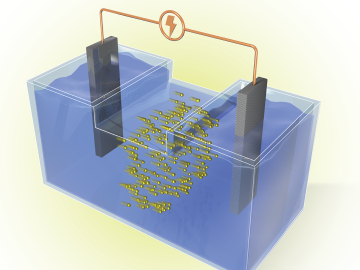
Researchers at ORNL are developing battery technologies to fight climate change in two ways, by expanding the use of renewable energy and capturing airborne carbon dioxide.

Jens Dilling has been named associate laboratory director for the Neutron Sciences Directorate at the Department of Energy’s Oak Ridge National Laboratory, effective April 1.

The Department of Energy’s Oak Ridge National Laboratory is providing national leadership in a new collaboration among five national laboratories to accelerate U.S. production of clean hydrogen fuel cells and electrolyzers.

Ateios Systems licensed an ORNL technology for solvent-free battery component production using electron curing. Through Innovation Crossroads, Ateios continues to work with ORNL to enable readiness for production-quality battery components.

Scientists from Stanford University and the Department of Energy’s Oak Ridge National Laboratory are turning air into fertilizer without leaving a carbon footprint. Their discovery could deliver a much-needed solution to help meet worldwide carbon-neutral goals by 2050.

Used lithium-ion batteries from cell phones, laptops and a growing number of electric vehicles are piling up, but options for recycling them remain limited mostly to burning or chemically dissolving shredded batteries.
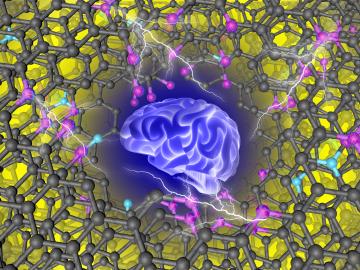
Guided by machine learning, chemists at ORNL designed a record-setting carbonaceous supercapacitor material that stores four times more energy than the best commercial material.
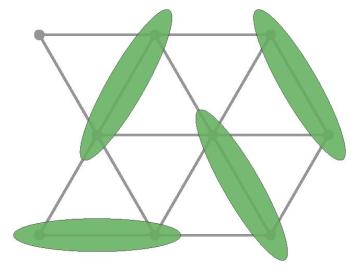
A team of researchers associated with the Quantum Science Center headquartered at the Department of Energy's Oak Ridge National Laboratory has confirmed the presence of quantum spin liquid behavior in a new material with a triangular lattice, KYbSe2.

Using neutrons to see the additive manufacturing process at the atomic level, scientists have shown that they can measure strain in a material as it evolves and track how atoms move in response to stress.

As current courses through a battery, its materials erode over time. Mechanical influences such as stress and strain affect this trajectory, although their impacts on battery efficacy and longevity are not fully understood.




