
Filter News
Area of Research
- Biology and Environment (14)
- Clean Energy (75)
- Climate and Environmental Systems (1)
- Computer Science (2)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Functional Materials for Energy (2)
- Fusion and Fission (4)
- Isotopes (1)
- Materials (34)
- Materials for Computing (4)
- National Security (2)
- Neutron Science (6)
- Supercomputing (8)
News Topics
- (-) Energy Storage (112)
- (-) Mercury (12)
- 3-D Printing/Advanced Manufacturing (128)
- Advanced Reactors (35)
- Artificial Intelligence (102)
- Big Data (62)
- Bioenergy (92)
- Biology (101)
- Biomedical (61)
- Biotechnology (24)
- Buildings (67)
- Chemical Sciences (73)
- Clean Water (31)
- Climate Change (106)
- Composites (30)
- Computer Science (199)
- Coronavirus (46)
- Critical Materials (29)
- Cybersecurity (35)
- Decarbonization (85)
- Education (5)
- Element Discovery (1)
- Emergency (2)
- Environment (201)
- Exascale Computing (44)
- Fossil Energy (6)
- Frontier (46)
- Fusion (59)
- Grid (67)
- High-Performance Computing (94)
- Hydropower (11)
- Irradiation (3)
- Isotopes (57)
- ITER (7)
- Machine Learning (51)
- Materials (149)
- Materials Science (149)
- Mathematics (10)
- Microelectronics (4)
- Microscopy (51)
- Molten Salt (9)
- Nanotechnology (60)
- National Security (73)
- Net Zero (14)
- Neutron Science (139)
- Nuclear Energy (111)
- Partnerships (51)
- Physics (64)
- Polymers (33)
- Quantum Computing (38)
- Quantum Science (72)
- Renewable Energy (2)
- Security (26)
- Simulation (53)
- Software (1)
- Space Exploration (25)
- Statistics (3)
- Summit (61)
- Sustainable Energy (130)
- Transformational Challenge Reactor (7)
- Transportation (99)
Media Contacts

To speed the arrival of the next-generation solid-state batteries that will power electric vehicles and other technologies, scientists led by ORNL advanced the development of flexible, durable sheets of electrolytes. They used a polymer to create a strong yet springy thin film that binds electrolytic particles and at least doubles energy storage.

At ORNL, a group of scientists used neutron scattering techniques to investigate a relatively new functional material called a Weyl semimetal. These Weyl fermions move very quickly in a material and can carry electrical charge at room temperature. Scientists think that Weyl semimetals, if used in future electronics, could allow electricity to flow more efficiently and enable more energy-efficient computers and other electronic devices.

Seven entrepreneurs comprise the next cohort of Innovation Crossroads, a DOE Lab-Embedded Entrepreneurship Program node based at ORNL. The program provides energy-related startup founders from across the nation with access to ORNL’s unique scientific resources and capabilities, as well as connect them with experts, mentors and networks to accelerate their efforts to take their world-changing ideas to the marketplace.

ORNL's Guang Yang and Andrew Westover have been selected to join the first cohort of DOE’s Advanced Research Projects Agency-Energy Inspiring Generations of New Innovators to Impact Technologies in Energy 2024 program. The program supports early career scientists and engineers in their work to convert disruptive ideas into impactful energy technologies.
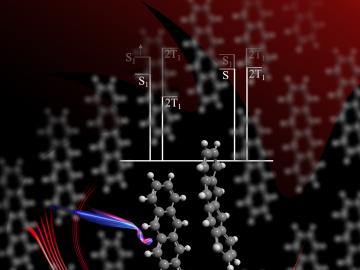
ORNL scientists develop a sample holder that tumbles powdered photochemical materials within a neutron beamline — exposing more of the material to light for increased photo-activation and better photochemistry data capture.
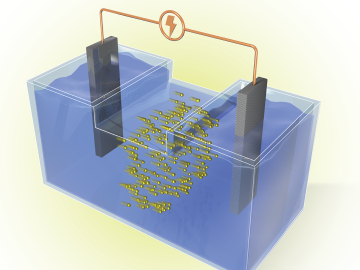
Researchers at ORNL are developing battery technologies to fight climate change in two ways, by expanding the use of renewable energy and capturing airborne carbon dioxide.

An international team using neutrons set the first benchmark (one nanosecond) for a polymer-electrolyte and lithium-salt mixture. Findings could produce safer, more powerful lithium batteries.

Shift Thermal, a member of Innovation Crossroads’ first cohort of fellows, is commercializing advanced ice thermal energy storage for HVAC, shifting the cooling process to be more sustainable, cost-effective and resilient. Shift Thermal wants to enable a lower-cost, more-efficient thermal energy storage method to provide long-duration resilient cooling when the electric grid is down.

The Department of Energy’s Oak Ridge National Laboratory is providing national leadership in a new collaboration among five national laboratories to accelerate U.S. production of clean hydrogen fuel cells and electrolyzers.

Chelsea Chen, a polymer physicist at ORNL, is studying ion transport in solid electrolytes that could help electric vehicle battery charges last longer.


