
Filter News
Area of Research
- Biology and Environment (95)
- Biology and Soft Matter (1)
- Clean Energy (118)
- Climate and Environmental Systems (5)
- Computational Engineering (1)
- Computer Science (3)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Functional Materials for Energy (2)
- Fusion and Fission (5)
- Isotopes (2)
- Materials (47)
- Materials for Computing (4)
- Mathematics (1)
- National Security (7)
- Neutron Science (14)
- Nuclear Science and Technology (1)
- Supercomputing (27)
News Topics
- (-) Energy Storage (112)
- (-) Environment (201)
- 3-D Printing/Advanced Manufacturing (128)
- Advanced Reactors (35)
- Artificial Intelligence (102)
- Big Data (62)
- Bioenergy (92)
- Biology (101)
- Biomedical (61)
- Biotechnology (24)
- Buildings (67)
- Chemical Sciences (73)
- Clean Water (31)
- Climate Change (106)
- Composites (30)
- Computer Science (199)
- Coronavirus (46)
- Critical Materials (29)
- Cybersecurity (35)
- Decarbonization (85)
- Education (5)
- Element Discovery (1)
- Emergency (2)
- Exascale Computing (44)
- Fossil Energy (6)
- Frontier (46)
- Fusion (59)
- Grid (67)
- High-Performance Computing (94)
- Hydropower (11)
- Irradiation (3)
- Isotopes (57)
- ITER (7)
- Machine Learning (51)
- Materials (149)
- Materials Science (149)
- Mathematics (10)
- Mercury (12)
- Microelectronics (4)
- Microscopy (51)
- Molten Salt (9)
- Nanotechnology (60)
- National Security (73)
- Net Zero (14)
- Neutron Science (139)
- Nuclear Energy (111)
- Partnerships (51)
- Physics (64)
- Polymers (33)
- Quantum Computing (38)
- Quantum Science (72)
- Renewable Energy (2)
- Security (26)
- Simulation (53)
- Software (1)
- Space Exploration (25)
- Statistics (3)
- Summit (61)
- Sustainable Energy (130)
- Transformational Challenge Reactor (7)
- Transportation (99)
Media Contacts

Daryl Yang is coupling his science and engineering expertise to devise new ways to measure significant changes going on in the Arctic, a region that’s warming nearly four times faster than other parts of the planet. The remote sensing technologies and modeling tools he develops and leverages for the Next-Generation Ecosystem Experiments in the Arctic project, or NGEE Arctic, help improve models of the ecosystem to better inform decision-making as the landscape changes.

Scientists using high-resolution aerial scans and computational modeling concluded that wildfires, storms and selective logging have become key drivers behind rainforest carbon emissions, outpacing clear-cutting practices.
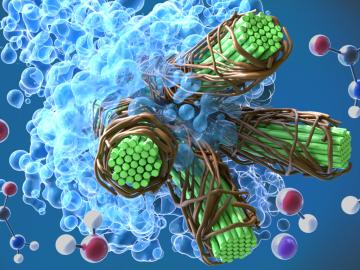
A team led by scientists at ORNL identified and demonstrated a method to process a plant-based material called nanocellulose that reduced energy needs by a whopping 21%, using simulations on the lab’s supercomputers and follow-on analysis.

To speed the arrival of the next-generation solid-state batteries that will power electric vehicles and other technologies, scientists led by ORNL advanced the development of flexible, durable sheets of electrolytes. They used a polymer to create a strong yet springy thin film that binds electrolytic particles and at least doubles energy storage.

A study found that beaches with manmade fortifications recover more slowly from hurricanes than natural beaches, losing more sand and vegetation. The researchers used satellite images and light detection and ranging data, or LIDAR, to measure elevation changes and vegetation coverage. Changes in elevation showed how much sand was depleted during the storm and how much sand returned throughout the following year.

At ORNL, a group of scientists used neutron scattering techniques to investigate a relatively new functional material called a Weyl semimetal. These Weyl fermions move very quickly in a material and can carry electrical charge at room temperature. Scientists think that Weyl semimetals, if used in future electronics, could allow electricity to flow more efficiently and enable more energy-efficient computers and other electronic devices.

Seven entrepreneurs comprise the next cohort of Innovation Crossroads, a DOE Lab-Embedded Entrepreneurship Program node based at ORNL. The program provides energy-related startup founders from across the nation with access to ORNL’s unique scientific resources and capabilities, as well as connect them with experts, mentors and networks to accelerate their efforts to take their world-changing ideas to the marketplace.

A research team led by the Department of Energy’s Oak Ridge National Laboratory demonstrated an effective and reliable new way to identify and quantify polyethylene glycols in various samples.

To better predict long-term flooding risk, scientists at the Department of Energy’s Oak Ridge National Laboratory developed a 3D modeling framework that captures the complex dynamics of water as it flows across the landscape. The framework seeks to provide valuable insights into which communities are most vulnerable as the climate changes, and was developed for a project that’s assessing climate risk and mitigation pathways for an urban area along the Southeast Texas coast.

In the wet, muddy places where America’s rivers and lands meet the sea, scientists from the Department of Energy’s Oak Ridge National Laboratory are unearthing clues to better understand how these vital landscapes are evolving under climate change.


