Filter News
Area of Research
- Advanced Manufacturing (1)
- Biological Systems (2)
- Biology and Environment (50)
- Clean Energy (71)
- Computer Science (2)
- Electricity and Smart Grid (3)
- Functional Materials for Energy (1)
- Fusion and Fission (4)
- Isotopes (1)
- Materials (17)
- Materials for Computing (2)
- National Security (19)
- Neutron Science (8)
- Nuclear Science and Technology (1)
- Quantum information Science (2)
- Sensors and Controls (2)
- Supercomputing (18)
News Topics
- (-) Bioenergy (86)
- (-) Grid (59)
- (-) Security (23)
- 3-D Printing/Advanced Manufacturing (115)
- Advanced Reactors (32)
- Artificial Intelligence (81)
- Big Data (49)
- Biology (93)
- Biomedical (56)
- Biotechnology (20)
- Buildings (53)
- Chemical Sciences (55)
- Clean Water (29)
- Climate Change (91)
- Composites (25)
- Computer Science (179)
- Coronavirus (46)
- Critical Materials (23)
- Cybersecurity (35)
- Decarbonization (70)
- Education (3)
- Element Discovery (1)
- Emergency (2)
- Energy Storage (106)
- Environment (188)
- Exascale Computing (33)
- Fossil Energy (4)
- Frontier (37)
- Fusion (51)
- High-Performance Computing (78)
- Hydropower (11)
- Irradiation (3)
- Isotopes (46)
- ITER (7)
- Machine Learning (43)
- Materials (137)
- Materials Science (130)
- Mathematics (6)
- Mercury (12)
- Microelectronics (2)
- Microscopy (50)
- Molten Salt (8)
- Nanotechnology (60)
- National Security (54)
- Net Zero (10)
- Neutron Science (127)
- Nuclear Energy (100)
- Partnerships (37)
- Physics (58)
- Polymers (31)
- Quantum Computing (28)
- Quantum Science (64)
- Renewable Energy (2)
- Simulation (41)
- Software (1)
- Space Exploration (24)
- Statistics (2)
- Summit (56)
- Sustainable Energy (115)
- Transformational Challenge Reactor (7)
- Transportation (92)
Media Contacts

ORNL researchers modeled how hurricane cloud cover would affect solar energy generation as a storm followed 10 possible trajectories over the Caribbean and Southern U.S.

ORNL’s Erin Webb is co-leading a new Circular Bioeconomy Systems Convergent Research Initiative focused on advancing production and use of renewable carbon from Tennessee to meet societal needs.
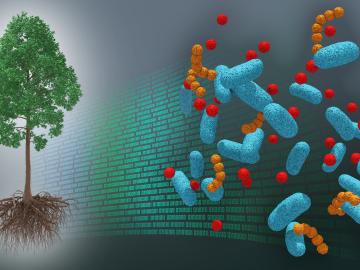
A first-ever dataset bridging molecular information about the poplar tree microbiome to ecosystem-level processes has been released by a team of DOE scientists led by ORNL. The project aims to inform research regarding how natural systems function, their vulnerability to a changing climate and ultimately how plants might be engineered for better performance as sources of bioenergy and natural carbon storage.

ORNL researchers are working to make EV charging more resilient by developing algorithms to deal with both internal and external triggers of charger failure. This will help charging stations remain available to traveling EV drivers, reducing range anxiety.

Alyssa Carrell started her science career studying the tallest inhabitants in the forest, but today is focused on some of its smallest — the microbial organisms that play an outsized role in plant health.
The United States could triple its current bioeconomy by producing more than 1 billion tons per year of plant-based biomass for renewable fuels, while meeting projected demands for food, feed, fiber, conventional forest products and exports, according to the DOE’s latest Billion-Ton Report led by ORNL.

An experiment by researchers at the Department of Energy’s Oak Ridge National Laboratory demonstrated advanced quantum-based cybersecurity can be realized in a deployed fiber link.

Scientists at ORNL are looking for a happy medium to enable the grid of the future, filling a gap between high and low voltages for power electronics technology that underpins the modern U.S. electric grid.

New computational framework speeds discovery of fungal metabolites, key to plant health and used in drug therapies and for other uses.

Scientists from more than a dozen institutions have completed a first-of-its-kind high-resolution assessment of carbon dioxide removal potential in the United States, charting a path to achieve a net-zero greenhouse gas economy by 2050.




