Filter News
Area of Research
- (-) Neutron Science (26)
- Advanced Manufacturing (22)
- Biological Systems (2)
- Biology and Environment (68)
- Building Technologies (2)
- Clean Energy (177)
- Climate and Environmental Systems (1)
- Computational Biology (1)
- Computational Engineering (3)
- Computer Science (16)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Functional Materials for Energy (2)
- Fusion and Fission (7)
- Fusion Energy (3)
- Isotopes (2)
- Materials (74)
- Materials for Computing (13)
- Mathematics (1)
- National Security (24)
- Nuclear Science and Technology (11)
- Quantum information Science (7)
- Supercomputing (104)
News Topics
- (-) 3-D Printing/Advanced Manufacturing (6)
- (-) Bioenergy (6)
- (-) Computer Science (13)
- (-) Energy Storage (6)
- Advanced Reactors (1)
- Artificial Intelligence (6)
- Big Data (2)
- Biology (5)
- Biomedical (11)
- Biotechnology (1)
- Chemical Sciences (2)
- Clean Water (2)
- Climate Change (1)
- Composites (1)
- Coronavirus (8)
- Cybersecurity (1)
- Decarbonization (2)
- Environment (8)
- Fossil Energy (1)
- Frontier (1)
- Fusion (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (14)
- Materials Science (23)
- Mathematics (1)
- Microscopy (3)
- Nanotechnology (10)
- National Security (2)
- Neutron Science (99)
- Nuclear Energy (3)
- Physics (9)
- Polymers (1)
- Quantum Computing (1)
- Quantum Science (7)
- Security (2)
- Space Exploration (3)
- Summit (6)
- Sustainable Energy (2)
- Transportation (5)
Media Contacts

Scientists at ORNL have developed 3D-printed collimator techniques that can be used to custom design collimators that better filter out noise during different types of neutron scattering experiments
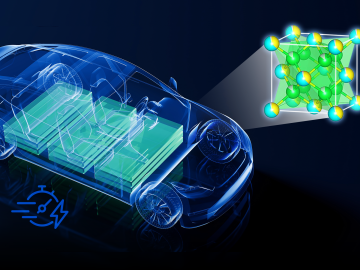
Currently, the biggest hurdle for electric vehicles, or EVs, is the development of advanced battery technology to extend driving range, safety and reliability.

The Department of Energy’s Office of Science has selected three ORNL research teams to receive funding through DOE’s new Biopreparedness Research Virtual Environment initiative.
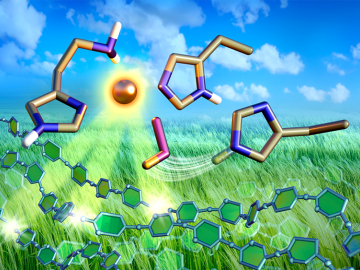
Nonfood, plant-based biofuels have potential as a green alternative to fossil fuels, but the enzymes required for production are too inefficient and costly to produce. However, new research is shining a light on enzymes from fungi that could make biofuels economically viable.

Researchers at ORNL have developed a new method for producing a key component of lithium-ion batteries. The result is a more affordable battery from a faster, less wasteful process that uses less toxic material.

Researchers at ORNL and the University of Tennessee, Knoxville, discovered a key material needed for fast-charging lithium-ion batteries. The commercially relevant approach opens a potential pathway to improve charging speeds for electric vehicles.

More than 50 current employees and recent retirees from ORNL received Department of Energy Secretary’s Honor Awards from Secretary Jennifer Granholm in January as part of project teams spanning the national laboratory system. The annual awards recognized 21 teams and three individuals for service and contributions to DOE’s mission and to the benefit of the nation.
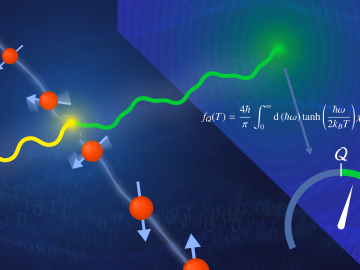
A team led by the U.S. Department of Energy’s Oak Ridge National Laboratory demonstrated the viability of a “quantum entanglement witness” capable of proving the presence of entanglement between magnetic particles, or spins, in a quantum material.
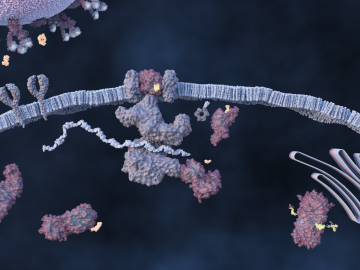
An ORNL-led team comprising researchers from multiple DOE national laboratories is using artificial intelligence and computational screening techniques – in combination with experimental validation – to identify and design five promising drug therapy approaches to target the SARS-CoV-2 virus.

At the Department of Energy’s Oak Ridge National Laboratory, scientists use artificial intelligence, or AI, to accelerate the discovery and development of materials for energy and information technologies.




