
Filter News
Area of Research
News Type
News Topics
- (-) Energy Storage (4)
- (-) Mercury (1)
- 3-D Printing/Advanced Manufacturing (12)
- Artificial Intelligence (2)
- Bioenergy (7)
- Biomedical (1)
- Biotechnology (1)
- Clean Water (4)
- Composites (2)
- Computer Science (7)
- Cybersecurity (1)
- Environment (12)
- Grid (2)
- Machine Learning (1)
- Materials Science (5)
- Microscopy (1)
- Nanotechnology (2)
- Neutron Science (2)
- Nuclear Energy (1)
- Polymers (1)
- Security (1)
- Space Exploration (1)
- Sustainable Energy (8)
- Transportation (10)
Media Contacts
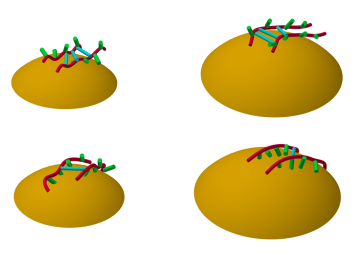
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

Sometimes solutions to the biggest problems can be found in the smallest details. The work of biochemist Alex Johs at Oak Ridge National Laboratory bears this out, as he focuses on understanding protein structures and molecular interactions to resolve complex global problems like the spread of mercury pollution in waterways and the food supply.
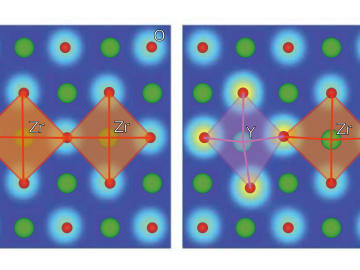
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

The use of lithium-ion batteries has surged in recent years, starting with electronics and expanding into many applications, including the growing electric and hybrid vehicle industry. But the technologies to optimize recycling of these batteries have not kept pace.
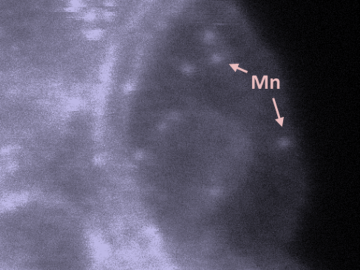
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.


