Filter Results
Related Organization
- Biological and Environmental Systems Science Directorate (23)
- Computing and Computational Sciences Directorate (35)
- Energy Science and Technology Directorate (217)
- Fusion and Fission Energy and Science Directorate (21)
- Information Technology Services Directorate (2)
- Isotope Science and Enrichment Directorate (6)
- National Security Sciences Directorate (17)
- Neutron Sciences Directorate (11)
- Physical Sciences Directorate
(128)
- User Facilities (27)
Researcher
- Sheng Dai
- Parans Paranthaman
- Bishnu Prasad Thapaliya
- Brian Post
- Zhenzhen Yang
- Craig A Bridges
- Shannon M Mahurin
- Sudarsanam Babu
- William Carter
- Alex Roschli
- Andrzej Nycz
- Blane Fillingim
- Chris Masuo
- Edgar Lara-Curzio
- Ilja Popovs
- Lauren Heinrich
- Li-Qi Qiu
- Luke Meyer
- Peeyush Nandwana
- Saurabh Prakash Pethe
- Thomas Feldhausen
- Tolga Aytug
- Uday Vaidya
- Yousub Lee
- Adam Stevens
- Ahmed Hassen
- Alexander I Wiechert
- Alexei P Sokolov
- Alex Walters
- Amy Elliott
- Anees Alnajjar
- Ben Lamm
- Beth L Armstrong
- Bruce Moyer
- Cameron Adkins
- Costas Tsouris
- Debangshu Mukherjee
- Eric Wolfe
- Erin Webb
- Evin Carter
- Frederic Vautard
- Gs Jung
- Gyoung Gug Jang
- Isha Bhandari
- Jayanthi Kumar
- Jeremy Malmstead
- Joshua Vaughan
- Kaustubh Mungale
- Kitty K Mccracken
- Liam White
- Md Inzamam Ul Haque
- Meghan Lamm
- Michael Borish
- Nageswara Rao
- Nidia Gallego
- Olga S Ovchinnikova
- Oluwafemi Oyedeji
- Peter Wang
- Phillip Halstenberg
- Radu Custelcean
- Ramanan Sankaran
- Rangasayee Kannan
- Roger G Miller
- Ryan Dehoff
- Santa Jansone-Popova
- Sarah Graham
- Shajjad Chowdhury
- Soydan Ozcan
- Subhamay Pramanik
- Tao Hong
- Tomonori Saito
- Tyler Smith
- Vimal Ramanuj
- Vlastimil Kunc
- Wenjun Ge
- William Peter
- Xianhui Zhao
- Yukinori Yamamoto

A novel strategy was developed to solve the limitations of the current sorbent systems in CO2 chemisorption in terms of energy consumption in CO2 release and improved CO2 uptake capacity.

This invention introduces a novel sintering approach to produce hard carbon with a finely tuned microstructure, derived from biomass and plastic waste.

Among the methods for point source carbon capture, the absorption of CO2 using aqueous amines (namely MEA) from the post-combustion gas stream is currently considered the most promising.

The increasing demand for high-purity lanthanides, essential for advanced technologies such as electronics, renewable energy, and medical applications, presents a significant challenge due to their similar chemical properties.

With the ever-growing reliance on batteries, the need for the chemicals and materials to produce these batteries is also growing accordingly. One area of critical concern is the need for high quality graphite to ensure adequate energy storage capacity and battery stability.

The use of biomass fiber reinforcement for polymer composite applications, like those in buildings or automotive, has expanded rapidly due to the low cost, high stiffness, and inherent renewability of these materials. Biomass are commonly disposed of as waste.
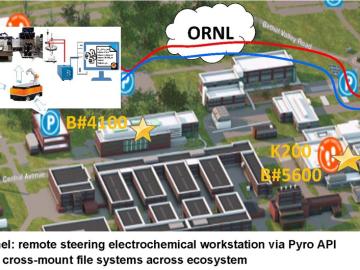
Electrochemistry synthesis and characterization testing typically occurs manually at a research facility.

A bonded carbon fiber monolith was made using a coal-based pitch precursor without a binder.

To develop efficient and stable liquid sorbents towards carbon capture, a series of functionalized ionic liquids were synthesized and studied in CO2 chemisorption via O–C bond formation.

This work seeks to alter the interface condition through thermal history modification, deposition energy density, and interface surface preparation to prevent interface cracking.

