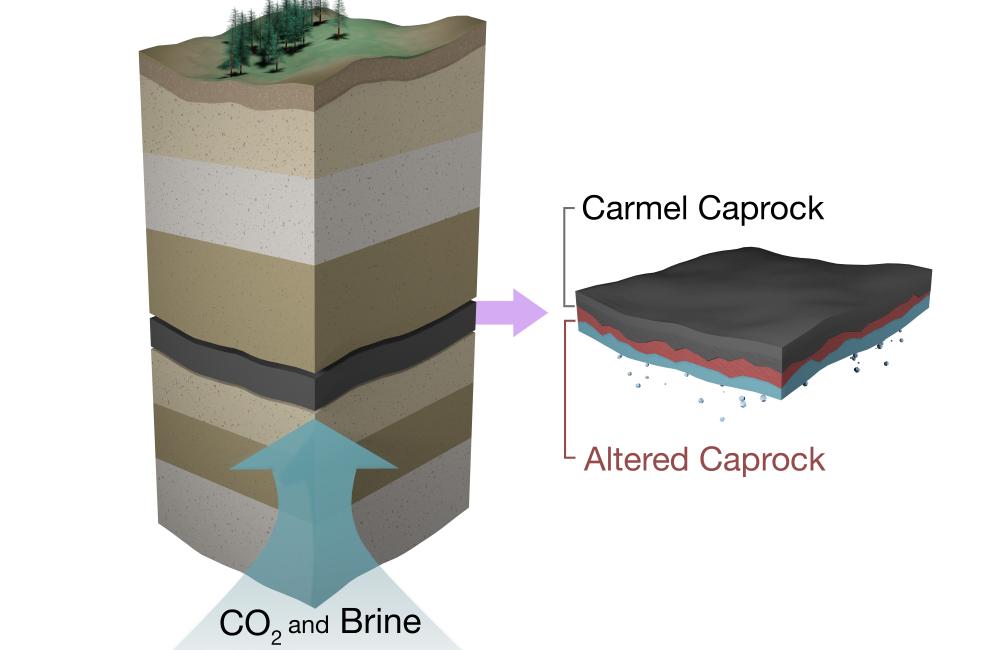
August 3, 2016 – Storing carbon dioxide for millennia in geologic formations relies on effective seals. Gernot Rother of Oak Ridge National Laboratory participated in analysis of neutron scattering data taken at the Jülich Centre for Neutron Science during a collaborative study led by Cambridge University and Shell Global Solutions. The investigation employed experiments and modeling to assess if a caprock trapped carbon dioxide supplied by an underlying aquifer. Researchers revealed a barrier-forming zone in which minerals dissolve, react with carbon dioxide and precipitate to clog pores. “Changes happening at the nanometer length scales that neutrons interrogate define how well caprock works as a barrier,” Rother said. “Coupled geochemical reactions and fluid flow phenomena in nanopores control the seal integrity. This particular caprock retains carbon dioxide far longer than expected.”
The Carmel caprock, which overlies an aquifer naturally charged with carbon dioxide, was analyzed with neutron scattering, revealing alteration only in the basal 7 cm and progression of caprock corrosion an order of magnitude slower than expected.
Media Contact
Dawn Levy
, Senior Science Writer and Communications Specialist, Physical Sciences Directorate
, 865.202.9465
|
LEVYD@ORNL.GOV