A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
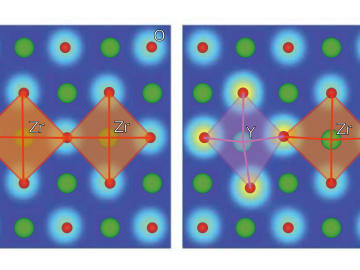
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice.

Lithium-ion batteries commonly used in consumer electronics are notorious for bursting into flame when damaged or improperly packaged. These incidents occasionally have grave consequences, including burns, house fires and at least one plane crash.

Oak Ridge National Laboratory scientists have developed a crucial component for a new kind of low-cost stationary battery system utilizing common materials and designed for grid-scale electricity storage.

The lighter wand for your gas BBQ, a submarine’s sonar device and the ultrasound machine at your doctor’s office all rely on piezoelectric materials, which turn mechanical stress into electrical energy, and vice versa.

Rechargeable batteries power everything from electric vehicles to wearable gadgets, but obstacles limit the creation of sleeker, longer-lasting and more efficient power sources.


