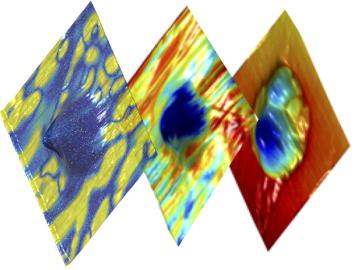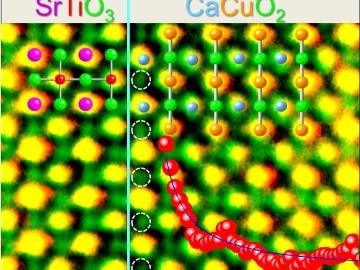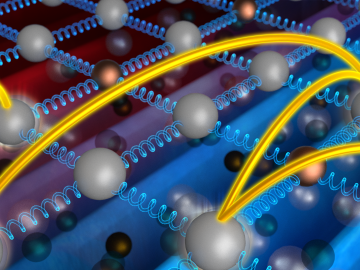
A discovery by scientists at the Department of Energy’s Oak Ridge National Laboratory supports a century-old theory by Albert Einstein that explains how heat moves through everything from travel mugs to engine parts.


Zili Wu of the Department of Energy’s Oak Ridge National Laboratory grew up on a farm in China’s heartland. He chose to leave it to catalyze a career in chemistry.
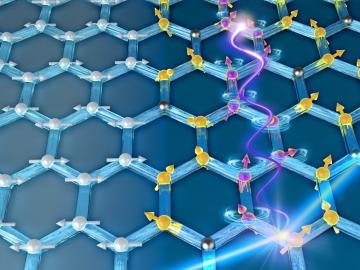
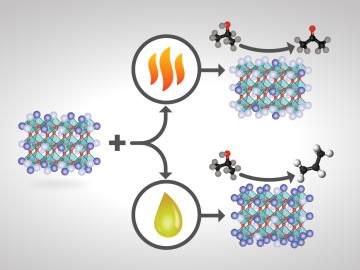
For some crystalline catalysts, what you see on the surface is not always what you get in the bulk, according to two studies led by the Department of Energy’s Oak Ridge National Laboratory. The investigators discovered that treating a complex
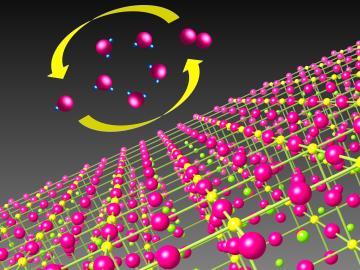
Catalysts make chemical reactions more likely to occur. In most cases, a catalyst that’s good at driving chemical reactions in one direction is bad at driving reactions in the opposite direction.