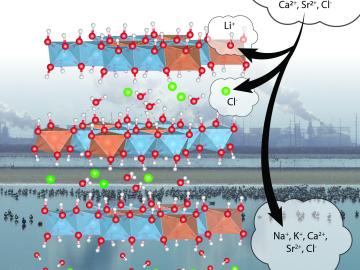
In the quest for domestic sources of lithium to meet growing demand for battery production, scientists at ORNL are advancing a sorbent that can be used to more efficiently recover the material from brine wastes at geothermal power plants.

In the quest for domestic sources of lithium to meet growing demand for battery production, scientists at ORNL are advancing a sorbent that can be used to more efficiently recover the material from brine wastes at geothermal power plants.

Two early career researchers at the Department of Energy's Oak Ridge National Laboratory have been included on the “Periodic Table of Younger Chemists” following an international competition conducted b
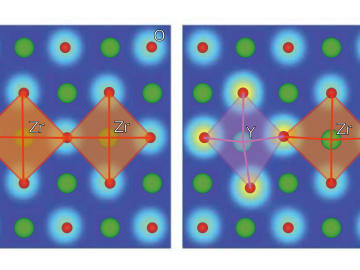
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice.
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.

