
Filter News
Area of Research
- (-) Biology and Soft Matter (5)
- (-) Chemistry and Physics at Interfaces (11)
- (-) Isotope Development and Production (3)
- Advanced Manufacturing (34)
- Biological Systems (18)
- Biology and Environment (179)
- Building Technologies (12)
- Chemical and Engineering Materials (4)
- Clean Energy (525)
- Climate and Environmental Systems (14)
- Computational Biology (6)
- Computational Chemistry (5)
- Computational Engineering (5)
- Computer Science (19)
- Data (1)
- Earth Sciences (1)
- Electricity and Smart Grid (3)
- Energy Frontier Research Centers (14)
- Energy Sciences (5)
- Fossil Energy (3)
- Fuel Cycle Science and Technology (3)
- Functional Materials for Energy (16)
- Fusion and Fission (55)
- Fusion Energy (19)
- Geographic Information Science and Technology (3)
- Isotopes (36)
- Materials (433)
- Materials Characterization (2)
- Materials for Computing (36)
- Materials Synthesis from Atoms to Systems (13)
- Materials Under Extremes (12)
- Mathematics (1)
- National Security (81)
- Neutron Data Analysis and Visualization (4)
- Neutron Science (192)
- Nuclear Science and Technology (74)
- Nuclear Systems Modeling, Simulation and Validation (3)
- Nuclear Systems Technology (1)
- Quantum Condensed Matter (4)
- Quantum information Science (9)
- Reactor Technology (1)
- Renewable Energy (4)
- Sensors and Controls (5)
- Supercomputing (313)
- Transportation Systems (11)
Media Contacts
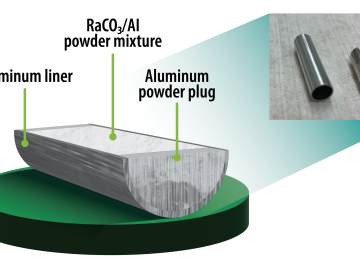
Oak Ridge National Laboratory researchers have developed a method to simplify one step of radioisotope production — and it’s faster and safer.

ORNL biogeochemist Elizabeth Herndon is working with colleagues to investigate a piece of the puzzle that has received little attention thus far: the role of manganese in the carbon cycle.

For many scientists and engineers, the first real test of their mettle comes not in a classroom, but in a lab or the field, where hands-on experience can teach volumes. For Susan Hogle, that hands-on experience just happened to be with material that was too hot to handle—literally....

With the production of 50 grams of plutonium-238, researchers at the Department of Energy’s Oak Ridge National Laboratory have restored a U.S. capability dormant for nearly 30 years and set the course to provide power for NASA and other missions.



From the bluebird painting propped against her office wall and the deer she mentions seeing outside her office window, Linda Lewis might be mistaken for a wildlife biologist at first glance. But rather than trailing animal tracks, Lewis, a researcher at the Department of Energy’s Oak Ridge National Laboratory, is more interested in marks left behind by humans.

With more than 30 patents, James Klett is no stranger to success, but perhaps the Oak Ridge National Laboratory researcher’s most noteworthy achievement didn’t start out so hot – or so it seemed at the time.

Less than 1 percent of Earth’s water is drinkable. Removing salt and other minerals from our biggest available source of water—seawater—may help satisfy a growing global population thirsty for fresh water for drinking, farming, transportation, heating, cooling and industry. But desalination is an energy-intensive process, which concerns those wanting to expand its application.

Graphene, a strong, lightweight carbon honeycombed structure that’s only one atom thick, holds great promise for energy research and development. Recently scientists with the Fluid Interface Reactions, Structures, and Transport (FIRST) Energy Frontier Research Center (EFRC), led by the US Department of Energy’s Oak Ridge National Laboratory, revealed graphene can serve as a proton-selective permeable membrane, providing a new basis for streamlined and more efficient energy technologies such as improved fuel cells.


