Filter News
Area of Research
- (-) Neutron Science (24)
- Advanced Manufacturing (3)
- Biology and Environment (102)
- Biology and Soft Matter (1)
- Building Technologies (1)
- Clean Energy (79)
- Climate and Environmental Systems (5)
- Computational Biology (1)
- Computational Engineering (3)
- Computer Science (15)
- Electricity and Smart Grid (1)
- Functional Materials for Energy (1)
- Fusion and Fission (25)
- Fusion Energy (13)
- Isotope Development and Production (1)
- Isotopes (26)
- Materials (49)
- Materials for Computing (8)
- Mathematics (1)
- National Security (34)
- Nuclear Science and Technology (20)
- Quantum information Science (6)
- Supercomputing (104)
News Topics
- (-) Computer Science (13)
- (-) Cybersecurity (1)
- (-) Environment (8)
- (-) Fusion (1)
- (-) Space Exploration (3)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Artificial Intelligence (6)
- Big Data (2)
- Bioenergy (6)
- Biology (5)
- Biomedical (11)
- Biotechnology (1)
- Chemical Sciences (2)
- Clean Water (2)
- Climate Change (1)
- Composites (1)
- Coronavirus (8)
- Decarbonization (2)
- Energy Storage (6)
- Fossil Energy (1)
- Frontier (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (14)
- Materials Science (23)
- Mathematics (1)
- Microscopy (3)
- Nanotechnology (10)
- National Security (2)
- Neutron Science (99)
- Nuclear Energy (3)
- Physics (9)
- Polymers (1)
- Quantum Computing (1)
- Quantum Science (7)
- Security (2)
- Summit (6)
- Sustainable Energy (2)
- Transportation (5)
Media Contacts

How did we get from stardust to where we are today? That’s the question NASA scientist Andrew Needham has pondered his entire career.

Natural gas furnaces not only heat your home, they also produce a lot of pollution. Even modern high-efficiency condensing furnaces produce significant amounts of corrosive acidic condensation and unhealthy levels of nitrogen oxides

Paul Langan will join ORNL in the spring as associate laboratory director for the Biological and Environmental Systems Science Directorate.

More than 50 current employees and recent retirees from ORNL received Department of Energy Secretary’s Honor Awards from Secretary Jennifer Granholm in January as part of project teams spanning the national laboratory system. The annual awards recognized 21 teams and three individuals for service and contributions to DOE’s mission and to the benefit of the nation.

Three ORNL scientists have been elected fellows of the American Association for the Advancement of Science, or AAAS, the world’s largest general scientific society and publisher of the Science family of journals.
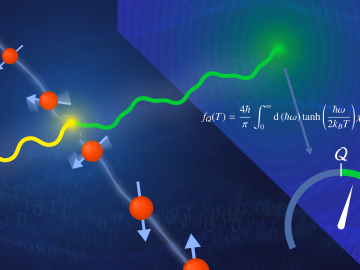
A team led by the U.S. Department of Energy’s Oak Ridge National Laboratory demonstrated the viability of a “quantum entanglement witness” capable of proving the presence of entanglement between magnetic particles, or spins, in a quantum material.
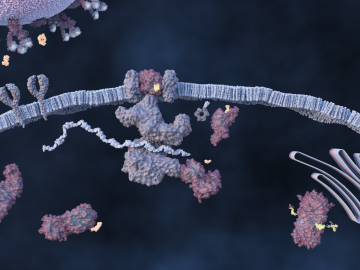
An ORNL-led team comprising researchers from multiple DOE national laboratories is using artificial intelligence and computational screening techniques – in combination with experimental validation – to identify and design five promising drug therapy approaches to target the SARS-CoV-2 virus.

Researchers from NASA’s Jet Propulsion Laboratory and Oak Ridge National Laboratory successfully created amorphous ice, similar to ice in interstellar space and on icy worlds in our solar system. They documented that its disordered atomic behavior is unlike any ice on Earth.

The Department of Energy’s Office of Science has selected five Oak Ridge National Laboratory scientists for Early Career Research Program awards.

At the Department of Energy’s Oak Ridge National Laboratory, scientists use artificial intelligence, or AI, to accelerate the discovery and development of materials for energy and information technologies.




