Filter News
Area of Research
News Topics
- (-) Transportation (6)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Bioenergy (1)
- Biomedical (2)
- Buildings (1)
- Chemical Sciences (4)
- Clean Water (1)
- Composites (4)
- Computer Science (1)
- Coronavirus (1)
- Critical Materials (5)
- Decarbonization (1)
- Energy Storage (7)
- Environment (1)
- Fusion (2)
- Isotopes (2)
- Materials (12)
- Materials Science (19)
- Microscopy (6)
- Molten Salt (1)
- Nanotechnology (8)
- Neutron Science (4)
- Nuclear Energy (3)
- Physics (2)
- Polymers (6)
- Quantum Computing (1)
- Quantum Science (1)
- Space Exploration (1)
- Sustainable Energy (3)
Media Contacts

Electric vehicles can drive longer distances if their lithium-ion batteries deliver more energy in a lighter package. A prime weight-loss candidate is the current collector, a component that often adds 10% to the weight of a battery cell without contributing energy.

ORNL scientists found that a small tweak created big performance improvements in a type of solid-state battery, a technology considered vital to broader electric vehicle adoption.

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.

Researchers at Oak Ridge National Laboratory proved that a certain class of ionic liquids, when mixed with commercially available oils, can make gears run more efficiently with less noise and better durability.
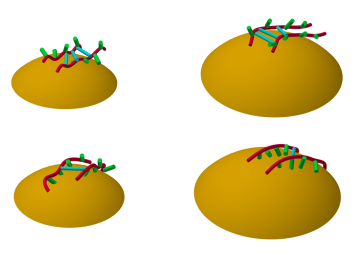
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
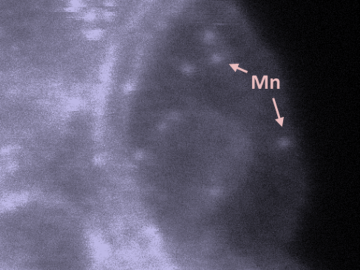
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.




