Filter News
Area of Research
News Topics
- (-) Advanced Reactors (1)
- (-) Energy Storage (7)
- (-) Grid (1)
- (-) Materials Science (20)
- (-) Nanotechnology (6)
- (-) Physics (4)
- 3-D Printing/Advanced Manufacturing (5)
- Artificial Intelligence (2)
- Big Data (1)
- Bioenergy (3)
- Biomedical (2)
- Clean Water (2)
- Composites (1)
- Computer Science (6)
- Cybersecurity (2)
- Environment (7)
- Fusion (2)
- Isotopes (1)
- Machine Learning (2)
- Microscopy (6)
- Molten Salt (1)
- Neutron Science (20)
- Nuclear Energy (7)
- Polymers (2)
- Quantum Science (3)
- Security (1)
- Sustainable Energy (5)
- Transportation (6)
Media Contacts

Oak Ridge National Laboratory has teamed with Cornell College and the University of Tennessee to study ways to repurpose waste soft drinks for carbon capture that could help cut carbon dioxide emissions.
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

A team led by scientists at the Department of Energy’s Oak Ridge National Laboratory explored how atomically thin two-dimensional (2D) crystals can grow over 3D objects and how the curvature of those objects can stretch and strain the

An ORNL-led team's observation of certain crystalline ice phases challenges accepted theories about super-cooled water and non-crystalline ice. Their findings, reported in the journal Nature, will also lead to better understanding of ice and its various phases found on other planets, moons and elsewhere in space.

OAK RIDGE, Tenn., May 7, 2019—Energy Secretary Rick Perry, Congressman Chuck Fleischmann and lab officials today broke ground on a multipurpose research facility that will provide state-of-the-art laboratory space
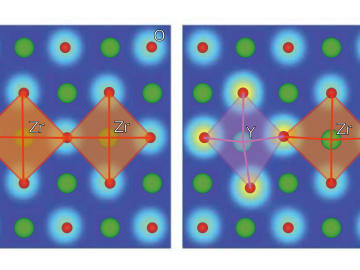
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

Kevin Field at the Department of Energy’s Oak Ridge National Laboratory synthesizes and scrutinizes materials for nuclear power systems that must perform safely and efficiently over decades of irradiation.
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and