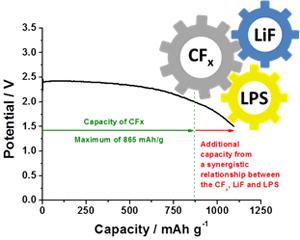
The novel Bi-functional electrolyte design yields additional capacity, resulting in a high energy density cathode that exceeds the theoretical capacity of conventional Li-CFx batteries.
An innovative design of a bi-functional electrolyte defies the theoretical maximum energy capacity of conventional lithium carbon fluoride (Li-CFx) batteries. This novel design has the potential of enabling the creation of long lasting batteries for implantable medical devices, wearable electronics and other applications. The discharge of CFx cathodes results in the formation of LiF that converts the solid electrolyte of Li3PS4 (LPS) to an electrochemical active component. This electrochemical activity is localized at the cathode, while the electrolyte remains stable at the anode. This selective stability enables the existence of a novel bi-functional electrolyte design. The additional capacity offered by the bi-functional electrolyte results in an observed capacity beyond the theoretical maximum for conventional Li-CFx cells. Additionally, amorphous LiF is formed in this solid-state cell. This material minimizes issues arising from solvents and crystalline LiF (volume expansion and heat generation) in conventional Li-CFx cells. A homogenous dispersion of electrode, electrolyte and the current collector results in a three-dimensional mixture delivering excellent performance at various discharge rates. The all-solid cell exhibits excellent stability and performance over a wide temperature range.
E. Rangasamy, J. Li, G. Sahu, N. J. Dudney, and C. Liang, “Pushing the Theoretical Limit of Li-CFx Batteries: A Tale of Bi-Functional Electrolyte,” Journal of the American Chemical Society, 2014. http://dx.doi.org/10.1021/ja5026358
For more information

